James J. Pestka
Degrees
PhD, Cornell University, 1979
BS, State University College at Buffalo, 1975
Professional Positions
Postdoctoral position at the University of Wisconsin at Madison, 1979-1982
Research Interests
Can omega-3 fatty acid consumption prevent environmental triggering of autoimmunity? Autoimmune disease (AD) affects over 25 million Americans, inflicting tremendous individual suffering and societal burdens. Managing AD today involves reducing disease symptoms in newly diagnosed individuals and preventing progression of established tissue damage to organs such as kidney. It is becoming increasing recognized that a hallmark of AD is ectopic lymphoid tissues (ELT) that morphologically and functionally resemble secondary lymphoid organs (e.g., lymph nodes) are likely to be a primary source of injurious autoreactive immune cells and autoantibodies. Existing AD therapeutics and emerging immunotherapies have multiple mechanisms of action, broadly encompassing non-specific immunosuppression, lymphocyte depletion, and neutralization of immune-stimulating cytokines and chemokines. These approaches have serious limitations: 1) unacceptable adverse effects; 2) resistance of ELT to their action; 3) inability to reverse immune-mediated damage; and 4) high costs.
While genetically inherited susceptibility (genome) is universally recognized as the primary predisposing factor for AD, lifetime environmental exposures (exposome) can either potentiate or attenuate AD manifestation. NIH expert committees have identified two exposome components critical to AD onset and progression: 1) toxic stressors and 2) dietary factors. Regarding toxic stressors, crystalline silica (cSiO2, quartz), the most abundant mineral on earth, is an environmental toxicant etiologically linked to human systemic lupus erythematosus (lupus) and other AD (rheumatoid arthritis, Sjögren’s syndrome, scleroderma, systemic vasculitis. An estimated 1.7 million Americans are exposed to respirable cSiO2 from manufacturing, mining, hydraulic fracturing, custodial service, farming, construction and military deployment. Consistent with epidemiological studies, we have found that airway exposure of cSiO2 to lupus-prone mice markedly decreases onset time and increases disease. As to dietary factors, animal and clinical studies suggest consuming ω-3 polyunsaturated fatty acids (PUFAs) found in fish oil, most notably docosahexaenoic acid (DHA) can resolve inflammation and might benefit individuals who suffer from AD including lupus. Our preclinical studies have now taken a step beyond AD symptom reduction to AD prevention by showing prevention of toxicant-triggered autoimmunity with dietary DHA might be achievable. ω-3s are the most widely consumed nutritional supplement after multivitamins, taken by ~30 million. DHA consumption by adults up to 5 g per day is considered safe . Given public acceptance and safety of DHA, discovering how supplementation with this ω-3 can protect against toxicant-triggered AD could have enormous benefits for American health.
It remains unclear how environmental toxicants like cSiO2 trigger initiation and flaring of ADs such as lupus and how DHA supplementation could be harnessed to block such environmental triggering. This critical knowledge gap places an unnecessary AD burden upon toxicant-exposed individuals that could otherwise be potentially mitigated by developing relatively low cost prophylactic approaches based on optimized ω-3 supplementation. Using lupus-prone NZBWF1 mice, we have established a model that permits dissection of the countervailing roles of cSiO2 (potentiation) and DHA (attenuation) in initiation and progression of autoimmunity. We identified several immunological endpoints (e.g., proinflammatory cytokines, chemokines, and B cell differentiation factors) active in lupus that are increased by intranasal cSiO2 exposure and -- much to our excitement -- are decreased dose-dependently by dietary DHA supplementation. Importantly, multiple endpoints inexpensively targeted by DHA are individually targeted by very expensive drugs approved either for or in clinical trials for lupus or other AD. An equally striking finding in our model was that cSiO2 exposure evoked formation of ectopic lymphoid tissue (ELT) in lungs that morphologically and functionally resemble secondary lymphoid organs (e.g., lymph nodes) and are likely to be a primary source of autoantibodies in bronchoalveolar fluid. Extraordinarily, dietary DHA supplementation dose-dependently blocked T and B cell infiltration and resultant ELT neogenesis in lung. This blockage may be intimately related to suppressed systemic autoimmunity and glomerulonephritis observed in NZBWF1 mice consuming DHA.
The goal of our research is to elucidate the early innate immune/inflammatory mechanisms by which cSiO2 elicits autoimmune effects and how DHA interferes with these events. This project will be the first step toward a mechanistic understanding of how dietary modulation of the lipidome could be used to prevent triggering of lupus. This knowledge is essential to develop practical, low-cost preventative strategies to help individuals environmentally exposed to cSiO2 and potentially other toxicants to reduce their risk for developing lupus or other AD. It will further be relevant as an intervention for stopping flare-ups and slowing progression of existing autoimmunity . We also anticipate our studies will reveal potential benefits of ω-3 consumption on other pulmonary inflammatory diseases, such as silicosis, another understudied area. New knowledge produced by this research project could help decrease morbidity and mortality associated with toxicant-triggered inflammation and autoimmunity, and reduce health care costs related to lupus and other AD. 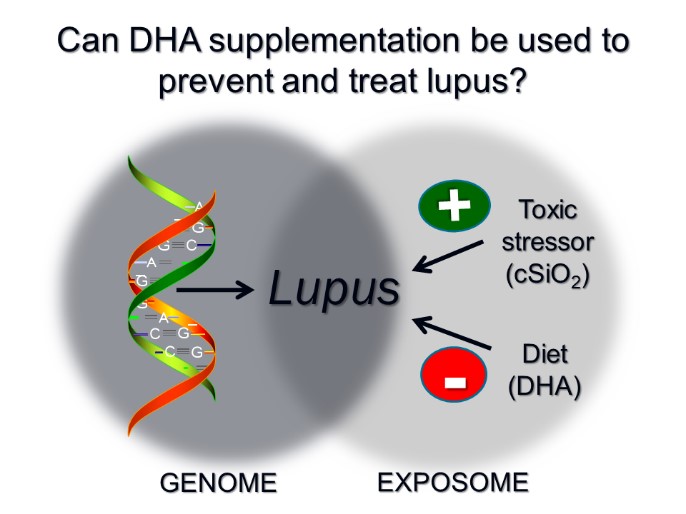
Publications
To view our citations, follow this Pubmed link to Dr. Pestka’s papers. A recent Web of Science Citation Report indicates his h-index is 55. We have published over 290 articles in several journals including Journal of Nutrition, Journal of Nutritional Biochemistry, PLOSone, Toxicological Sciences, Toxicology and Applied Pharmacology, Toxicology, Journal of Food Protection. Dr. Pestka ranks first in the number of Papers among 9,727 authors in mycotoxin field (read more)
Awards
Dr. Pestka's honors include: Robert and Carol Deibel Endowed Professorship for Lupus Research; Promotion to University Distinguished Professorship at MSU in 2011 (see more); Distinguished Faculty Award, MSU 1999; Distinguished Faculty Award, MSU College of Agriculture and Natural Resources (CANR), 1996; Elected Fellow of Academy of Microbiology, 1995; Elected to Phi Kappa Phi Honor Society, 1995; Harvey W. Wiley Award, Association of Official Analytical Chemists, 1993; Junior Faculty Meritorious Research Award, Michigan State University Chapter of Sigma Xi, 1992; Carl G. Smith Award, MSU CANR, 1992; Samuel Cate Prescott Award, Institute of Food Technologists, 1988; Teacher-Scholar Award, MSU, 1987; Wise and Helen Burroughs Lectureship, Nutritional Sciences Council of Iowa State University, 1986; Young Faculty International Travel Award, MSU CANR, 1986; Dean's Award for Outstanding Teaching in Microbiology, New York State College of Agriculture and Life Sciences, 1978; Presented for distinguished performance as Teaching Assistant in the Dept. of Microbiology. Undergraduate: New York State Regents Scholarship, Magna Cum Laude.
Instructional Activities
Dr. Pestka teaches Advanced Food Toxicology (FSC 807). This graduate course focuses on the major classes of toxic chemicals in food relative to occurrence, metabolism, molecular/cellular/tissue targets, risk assessment and regulation. The course will involve lectures with accompanying readings from texts and recent reviews. In addition, there will be discussions of recent peer-reviewed papers dealing with the occurrence, toxicology and safety assessment of major classes of toxic foodborne chemicals. Current lecture topics will include: 1) food toxicology principles , 2) detection of toxic chemicals in food, 3)absorption, distribution, metabolism and excretion of toxic foodborne chemicals, 4)safety evaluation of chemicals and the “FDA Red Book”, 5)food additives and the GRAS list, 6)toxicants formed during food processing/packaging, 7)anthropogenic contaminants (e.g. pollutants, pesticides), 8)biological toxins (microbial, plant, animal), 9) nutrients and functional foods, 10)food allergens and 11)emerging Issues
Other connections to Dr. Pestka
Department of Microbiology and Molecular Genetics
Center for Integrative Toxicology
MSU Scholars
Related Work
-
MSU researcher instrumental in gaining insight into rising concern over air pollution
Published on July 19, 2023
-
Michigan State University graduate student honored for lupus research
Published on September 17, 2019
-
Gift of $1.5 million supports lupus research
Published on May 14, 2018
-
Lupus: Preventing onset from an environmental trigger
Published on February 22, 2018
-
AgBioResearcher Top Publisher of Mycotoxins Papers
Published on August 20, 2009









 Print
Print Email
Email