Phytophthora root rot update
Phytophthora (Phytophthora nicotianae and Phytophthora drechsleri are examples) can be found in floriculture crops and can cause root, crown, and foliar blights. Losses can be especially severe in greenhouses and production fields where warm temperatures and ample water favor epidemics. Recirculating irrigation water can enhance the spread of Phytophthora.
Phytophthora is difficult to control because it produces several different spores that can cause disease. Thick-walled oospores survive between crops on plant containers, benches, floors, and in potting media or soil. Other spores include lemon-shaped sporangia, thick-walled chlamydospores, and swimming zoospores. Water is important for disease and under warm, wet conditions many spores develop on infected plants and lead to a rapid build-up of disease and spread in a short period of time.
Once an epidemic has developed in a production site it is often difficult to determine how the Phytophthora was introduced, how it is spreading, and if it is surviving from year to year. Using genetic tools, research conducted at MSU showed that Phytophthora spread within the greenhouse can occur from sporangia that can travel through the air or zoospores that spread through the water. Recent research also shows how Phytophthora may spread among producers. For instance, snapdragon producers at two locations purchased plugs from the same supplier. The Phytophthora from the two locations was identical. While it is possible that the disease was introduced to the locations via infected plants, it is also possible that the Phytophthora was already established at these production sites. The Phytophthora recovered from snapdragons was identical to the Phytophthora collected from the same facility in a previous year indicating the potential for this pathogen to survive even with a fallow period and treatment with a fumigant (methyl bromide/chloropicrin).
In another example, verbena propagated at one greenhouse via cuttings was sold to two other greenhouses for finishing. The finding that the Phytophthora from all three locations were genetically identical suggests that infected plants could have spread P. nicotianae from the propagator to the two growers.
Controlling the spread of Phytophthora spp. within and among production facilities can be difficult and there are two major challenges. First, Phytophthora must be kept out of the production site. This is particularly difficult with floriculture crops because of the widespread distribution of pre-finished plants. Also, plants may not exhibit obvious symptoms until the infection is well established or the plant becomes stressed (e.g., over- or under-watering). Infected plants treated with fungicides may appear healthy until the fungicides wear off and Phytophthora increases. The second challenge is eradicating Phytophthora once it has been introduced. Removing visibly diseased plants will not prevent spore production and spread from plants showing few if any symptoms. Sanitation can limit disease and includes removing plant debris, disinfecting pots, and production surfaces. Routinely treating plants with fungicides including mefenoxam (Subdue MAXX) can be helpful. However, Phytophthora can develop resistance to these fungicides and new management strategies and tools are needed.
Evaluation of registered and unregistered fungicides in managing Phytophthora root rot of poinsettia
Cuttings of poinsettia ‘Freedom Red’ rooted in oasis cubes were transplanted into four-inch pots containing a soilless medium on June 19. Inoculum was prepared by growing an isolate of Phytophthora drechsleri on ¼-strength V8 agar for one week. Two 0.48-inch plugs of P. drechsleri were placed on each side of the oasis cube of each rooted cutting during transplanting into four-inch plastic pots on June 19. Six one-plant replicates per treatment were arranged in a completely randomized design. Immediately after transplanting, treatments were applied as a drench. Treatments were applied again on July 11 and 25. Plants were fertilized twice weekly with 200 ppm Peter’s 20-20-20 liquid fertilizer, and were grown at 85ºF day temperature, 67ºF night temperature. Plant health (1-5; 1=healthy, 2=chlorosis/stunting, 3=minor wilting, 4=severe wilting, 5=plant death), and death (%) were assessed on June 29, July 18, and August 1.
Disease pressure was severe with 66.7 percent of the untreated inoculated plants dead with the remaining alive plants severely stunted. Adorn 4FL (both rates) and commercial standard Subdue MAXX were the only treatments that resulted in plants showing no symptoms from Phytophthora and received plant health ratings of 1.0 (1=healthy, 5=plant death). Alude significantly reduced infection compared to the untreated inoculated while ZeroTol had 100 percent plant death by the second rating date. A high and low rate of the unregistered fungicide Cyazofamid, Heritage, and Adorn were included in this trial with no significant differences between the rates for each treatment on the last rating date. Strobilurin products Heritage and Insignia were not effective in controlling infection and resulted in plant health and death (percentage) similar to that of the untreated inoculated. No phytotoxicity was observed on any of the treated plants in this trial.
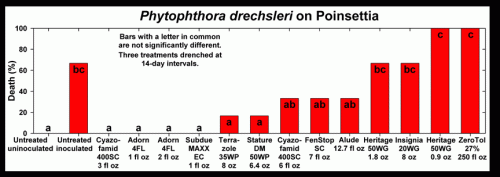
Figure 1.
Acknowledgement for resistance management and disease update articles
This research was funded in part by a Cooperative Agreement #59-1907-5-553 with the Agricultural Research Service and the American Floral Endowment.



 Print
Print Email
Email

