Sensitivity of American brown rot to sterol inhibitor fungicides in 2008
Editor’s note: This article is from the archives of the MSU Crop Advisory Team Alerts. Check the label of any pesticide referenced to ensure your use is included.
American brown rot ( AMERICAN BROWN ROT) is caused by the ascomycete fungus Monilinia fructicola, and is an important pathogen on cherry (particularly sweet cherry varieties), peach, apricot, nectarine, and plum. The fungus attacks fruit, blossoms, spurs and shoots with ideal infection conditions initiating epidemic inoculum levels in as little as 24 hours. Brown rot causes fruit rot before and after harvest, greatly reducing the quality and quantity of the yield, particularly in heavily bunching sweet cherry varieties.
During the 2008 growing season many factors contributed to the high level of brown rot observed in sweet cherry orchards around the state. Pollination problems, unusually long retention of unfertilized cherries, and wind and hail damage combined with ideal conditions for disease development leading up to harvest creating epidemic levels of brown rot infection in many sweet cherry blocks around the state. Due to the high levels of brown rot, the efficacy of sterol inhibitor fungicides, such as Indar, Orbit and Elite, was called into question. Field trials testing Elite and Indar were conducted at the Northwest Michigan Horticultural Research Station in Traverse City, Michigan during 2008. Disease pressure was extraordinarily high because we inoculated trees with the brown rot pathogen. Despite this high pressure, Indar adequately suppressed brown rot (Table 1). Elite was not as effective as in previous trials and will be tested again in 2009 to confirm these observations.
Table 1. 2008 M.fruticola fungicide efficacy data
In addition to field efficacy trials, in vitro sensitivity of M. fructicola to Indar and Orbit was assessed in 2008. Twenty-one sweet and nine tart cherry orchards were screened, from northern Leelanau County south to Benton Harbor. Thirty M. fructicola samples were collected at each orchard and were then single-spore isolated in the lab. Fungicide resistance studies use reduced mycelial growth to measure sensitivity. In this case, we examined growth of the fungi on agar amended with sterol inhibitors, relative to normal growth of the fungi in the absence of the inhibitor. Propiconazole concentrations used in our tests were based on a discriminatory dose (0.3µg/ml a.i. propiconazole) established in peaches (Schnabel, et al. 2004), a stone fruit generally less susceptible to brown rot. This discriminatory dose defines sensitive M. fructicola isolates as those that do not grow on agar amended with 0.3µg/ml a.i. propiconazole (the discriminatory dose). Resistant isolates are defined as those which have a relative growth of greater than or equal to 50 percent on agar amended with the discriminatory dose of 0.3µg/ml a.i. propiconazole, compared to a control. In the case of Indar, the discriminatory dose of 0.1µg/ml a.i. of fenbuconazole was based on a discriminatory dose that we established in 2007 by sampling an abandoned Michigan orchard, representing a fungal population with minimal exposure to sterol inhibitor fungicides.
None of the orchards screened confirm resistance to propiconazole (Orbit) but many orchards, particularly sweet cherry orchards are approaching 50 percent relative growth, characteristic of resistant M. fructicola isolates in peaches (Figure A). Currently, this screening process cannot confirm or reject the presence of fenbuconazole (Indar) resistance as practical field resistance has not been conclusively proven (Figure B). Indar sensitivity levels can be used to gauge changes in sensitivity over time and to compare the average sensitivity of orchards based on the history of sterol inhibitor use.
Figure A.
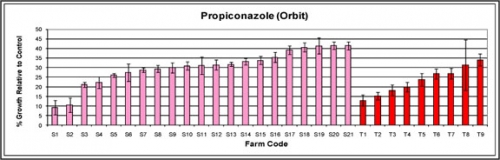
Average relative growth of Monilinia fructicola isolates in the presence of propiconazole (Orbit) based on orchard site. Pink bars indicate samples collected from sweet cherry orchards, red bars indicate samples collected from tart cherry orchards. Greater than or equal to 50 percent relative growth would be indicative of propiconazole resistance.
Figure B.
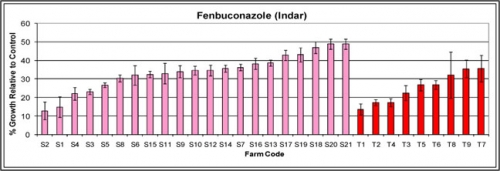
Average relative growth of Monilinia fructicola isolates in the presence of fenbuconazole (Indar) based on orchard site. Pink bars indicate samples collected from sweet cherry orchards, red bars indicate samples collected from tart cherry orchards.
Interesting patterns emerge from the data when orchards are sorted based on past sterol inhibitor use. There are few sites available with limited or no history of sterol inhibitor use, so sampling is limited to organic/organic-transitional sites, and abandoned blocks. Despite the small sample size, significant differences in M. fructicola sensitivity exist based on sterol inhibitor history (Figure C). These data point to sterol inhibitor fungicides having a definite effect on M. fructicola population sensitivity, the same pattern which was confirmed in a 2007 survey of brown rot.
Figure C.
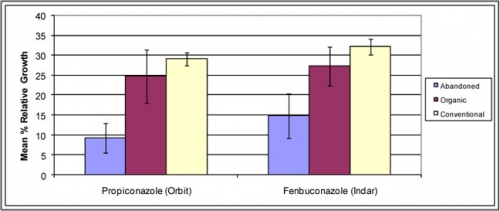
Monilinia fructicola sterol inhibitor-sensitivity based on history of use in abandoned, organic and conventional orchards.
The results of the in vitro, M. fructicola inhibitor sensitivity trial produced two indicators that sterol inhibitors are influencing shifts in the fungal populations. First, there is an overall variability in M. fructicola sensitivity, an indicator that the population is being affected by the fungicide (Figures A and B). Second, there is a significant difference in the population’s sensitivity based on the past exposure of the fungi to the fungicide (Figure C).
What methods can be used to limit further shifting in the brown rot populations that will lead to resistance to sterol inhibitors? Tank mixing and fungicide rotation are a must to preserve SI efficacy for as long as possible. Full rates and good coverage are also very important. Iprodione (Rovral® 50WP) may be applied for the spring, blossom-blight application to help limit the number of sterol inhibitor sprays used in a season. Iprodione is a protectant fungicide that affects the fungus differently than sterol inhibitors and may help reduce the speed at which sterol inhibitor resistance is developing. Pyraclostrobin and boscalid mixtures (Pristine®) and trifloxistrobin (Gem) are also rated as providing good control of American brown rot under lower disease pressure and can be utilized as a rotational partner during some seasons. We are also awaiting word on a special Section 24(c) label for Indar which will increase the allowable rate used in orchards from 6 fl oz per acre to 6-12 fl oz per acre. Since differences in sterol inhibitor sensitivities are quantitative in the brown rot population, increasing the rate of Indar should be effective in controlling strains with shifted phenotypes.
The most important factor in limiting further shifting in brown rot populations is to effectively kill the fungus in orchards and limit exposure to sterol inhibitors. This can be best accomplished as stated above by rotating modes of action (especially to Iprodione during bloom). In addition, increasing the rate of Indar initially to at least 8 fl oz per acre would increase the effectiveness of the best tool for killing American brown rot fungi. We will pass word along of the Section 24(c) label when it becomes available.



 Print
Print Email
Email

