
Bulletin E-3412 Introduction to Fertilizer Planning
DOWNLOADJanuary 8, 2021 - Jon LaPorte
As a young or beginning farmer, you may be raising crops to reduce feed costs or to sell on the market. One of the important decisions in crop production is deciding how much fertilizer you should apply. Fertilizer application focuses on meeting the nutrient needs of the crop you’ll grow. Too much fertilizer and potential profits are reduced. Too little fertilizer and there is not enough production. To meet the nutrient needs and produce a profitable crop, you’ll need to create a fertilizer plan.
This publication explores the process of creating a fertilizer plan. This includes spending some time understanding the soil and the available nutrients within it. You’ll also learn about the available forms of fertilizer and the methods of applying them to the soil. Using the information collected from both the soil and the available fertilizer options, you’ll begin to create a fertilizer plan for the farm. This includes understanding what the costs are in the plan and their impacts on farm revenue.
Many factors go into fertilizer planning. Understanding the soil is the first consideration since this is where fertilizer will be applied. The properties and characteristics of the soil in your field can greatly affect the types of fertilizers and application methods you will want to use
Soil tests and their results are important to knowing what nutrients are already present and if they are available to the intended crop. Soil characteristics and the availability of nutrients is crucial information as you start to think about the amount of nutrients needed overall.
The amount of nutrients needed is influenced by what the yield goals are and whether the soil needs to be built up or if nutrient needs should only consider crop removal rates. Finally, how these nutrients will be applied completes the decision factors needed for a successful and profitable crop. You can use many types of fertilizer. When you apply them is critical to a successful plan.
Understanding the Soil
You will get your plan started by understanding the soil that you’re working with in the fields.
Soil is made up of soil particles, air, and water. Ideally, soil should be filled with 25% water and 25% air and the other 50% with soil particles (see Figure 1). This maximizes the availability of nutrients to roots and keeps a healthy environment for soil microbes that help with nutrient uptake.

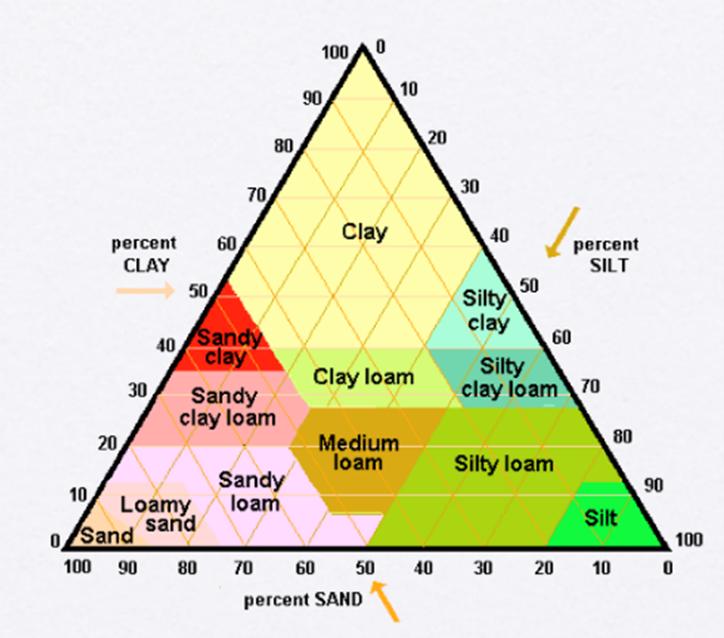
The type of particles often varies by the amount of sand, silt, or clay (see Figure 2). Depending on the amount of each type of particles you have in your soil, the ability to interact with nutrients will be different from field to field. Knowing the soil type helps you understand what fertilizer options and strategies are best. For more information on the soil types in your field, visit the United States Department of Agriculture Web Soil Survey (https://websoilsurvey.sc.egov.usda.gov/App/HomePage.htm).
Use the pH scale to guide you toward that chemical balance and achieve ideal growing conditions for the intended crop. The optimum pH level for corn, soybeans, and wheat is 6.5. pH at slightly below or above these levels is acceptable, but 6.5 is the desired target. A level of 6.5 is optimal for these crops because it allows them access to those nutrients needed in larger quantities (see Figure 3).
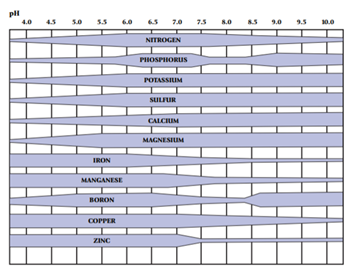
Depending on the intended crop, the optimal levels may be different, such as blueberries that prefer a pH of 5.5. In the case of most row crops, such as corn, soybeans, and wheat, a pH of 6.5 allows access to several important nutrients. This includes nitrogen, phosphorus, potassium, sulfur, calcium, and magnesium.
To identify soil characteristics and the available nutrients present, routine soil testing is necessary. Ideally, you should test soil every three years. Soil tests tell where the pH levels are and help track them over time. If pH levels are low, a soil amendment called lime may be needed to bring pH back up to optimum levels. The Michigan State University (MSU) Extension bulletin Lime for Michigan Soils (E-471) (https://www.canr.msu.edu/uploads/resources/pdfs/lime-for-mi-soils-e-471.pdf) provides guidance on using lime to correct pH levels.
Soil tests also help to track the amount of nutrients available and if those levels are increasing or decreasing over time. If nutrient levels are low, MSU Extension recommends using build-up rates of fertilizer to bring nutrient levels up to a more sufficient level. If they are high, MSU Extension recommends applying only what the crop will remove at harvest.
To understand the amount of nutrients to apply to the soil, consider the farm’s yield goals. Yield goals have several determining factors. They include the soil type, the cation exchange capacity (CEC) level, the availability of water throughout the growing season, and the experience of the producer growing the crop.
Cation Exchange Capacity
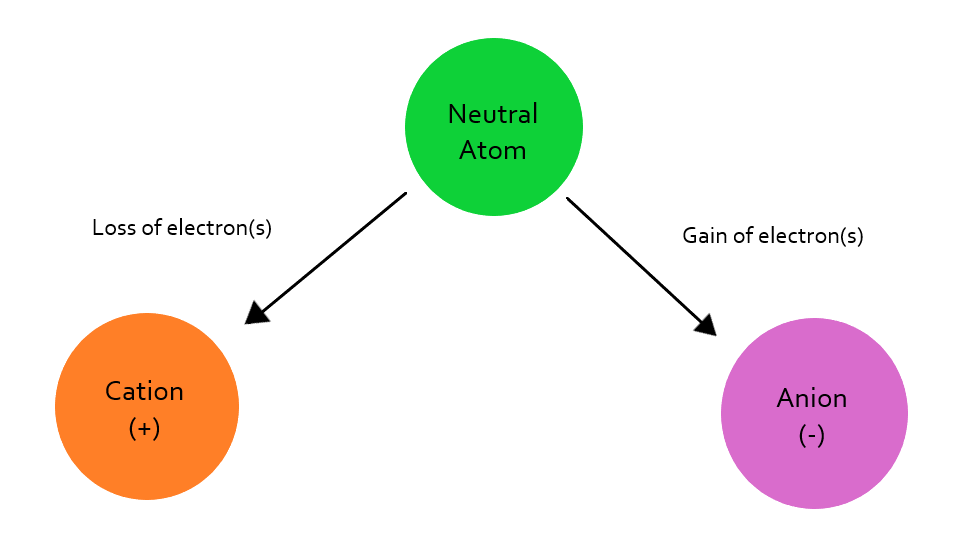
While all of these factors are important, the CEC is perhaps the most important in determining a yield goal. CEC is a measure of the soil’s ability to hold positively charged ions. All nutrients are made up of ions. The ions with a positive charge are called cations and the ions with negative charges are called anions (see Figure 4). Soils can readily hold cations, but they hold few anions. The higher the CEC level, the more positively charged nutrients, or cations, the soil can hold.
The soil’s structure and stability when growing a crop influences the CEC. This is a concern in areas of severe weather. The CEC influences nutrient availability, pH levels, and even how the soil will react to fertilizers. For example, an old standby used by producers is to take the CEC level and multiply by two, giving an indication of the nitrogen holding capacity of the soil.
Consider the types of nitrogen fertilizers that can be applied to the soil (see Table 1). Ammonium sulfate and anhydrous ammonia contain ammonium ions, which are positively charged cations. The soil can hold on to these nutrients for a longer period of time without the immediate concern of nutrient leaching. Nutrient leach is the downward movement of dissolved nutrients in the soil with gradually moving water.
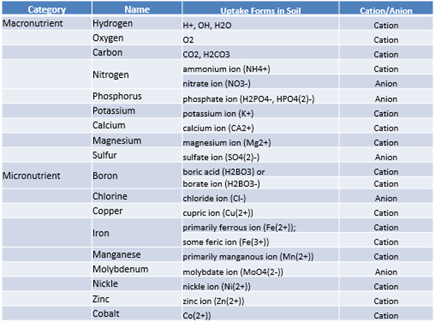
On the other hand, nitrogen fertilizers such as urea ammonium nitrate (UAN) are mostly made up of nitrate ions, which are negatively charged anions. The soil does not hold on to anions for a significant amount of time, leading to more concerns of nutrient leaching with these types of products.
The CEC is also an indicator of the soils ability to hold water. Water, or H20, is made up of ions, too. Soils with higher amounts of silt and clay have higher CEC levels than sandy soils, so they can hold on to larger amounts of water and nutrients. The higher the CEC levels, the greater amount of water-holding capacity the soils have. Typically, CEC levels of 11 to 50 retain more water than levels below 11.
Rate of Nutrients to Apply
Equipped with the soil’s ability to hold on to water and nutrients, as well as soil test results for the farm, the next consideration is the rate of nutrients to apply. There are several methods of determining nutrient amounts to apply, including build-up and maintenance or nutrient removal only. Another option is the Marginal Return to Nitrogen (MRTN) method that combines nutrient needs and economics. For more information on the MRTN method, review the MSU Extension article “Corn Nitrogen Recommendations” (https://www.canr.msu.edu/news/corn_nitrogen_recommendations).
A build-up and maintenance approach involves applying enough fertilizer to keep the soil at healthy levels. The application of nutrients will meet the farm’s yield goals, but often requires more fertilizer than focusing only on nutrient removal. However, if soil levels are high enough, this method can apply amounts less than nutrient removal.
The other method to consider is nutrient removal only. This method applies only the rate of fertilizer needed to replace what nutrients the crop will remove at harvest. Table 2 is adapted from the MSU Extension bulletin Nutrient Recommendations for Field Crops in Michigan (E-2904) (https://soils.msu.edu/wp-content/uploads/2014/06/MSU-Nutrient-recomdns-field-crops-E-2904.pdf). It highlights that crop removal depends on how many bushels or tons will be removed from the field. Each bushel or ton removes a certain amount of nutrients. The more bushels or tons produced, the more nutrients will be removed.
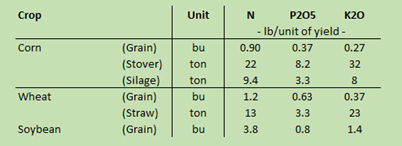
Since production numbers won’t be known until harvest, you’ll need to finalize the farm’s yield goal to fully use the table as part of your fertilizer planning. Look at an example using a field of corn.
To find the amount of nutrients that will need to be replaced, you will need to calculate the yield goal for the corn crop. This means locating information on the soil type, the CEC level, the availability of water throughout the growing season, and the experience of the producer growing the crop.
|
Table 3. Example Corn Farm |
|
|
Soil Type: |
Sandy Loam |
|
Cation Exchange Capacity (CEC) Level: |
5 |
|
Availability to Water: |
No irrigation – dryland field |
|
Grower Experience: |
10 years of raising corn |
|
Expected Yield Goal: 150 bushels per acre |
|
Information on the example farm in Table 3 reveals that the field has a soil type of sandy loam and CEC levels from the soil test results are below 11. These results indicate there is not a great deal of water or nutrient-holding capacity in the soil. The field is also considered “dryland” as it does not have an irrigation system, which means its availability to water is dependent solely on rainfall. Lastly, the producer has roughly 10 years of experience raising corn and understands how it reacts in this type of environment. The producer calculates an expected yield goal of 150 bushels per acre for this example farm.
Table 2 (the previous nutrient removal chart) shows that every bushel expected at harvest will remove 0.9 lbs of nitrogen, 0.37 lbs of phosphorus, and 0.27 lbs of potassium. Those nutrient removal values should be multiplied by the yield goal of 150 bushels. This results in 135 lbs of nitrogen, 55 lbs of phosphorus, and 40 lbs of potassium that will need to be replaced per acre to maintain the soil at current nutrient levels.
The fertilizer needs of this example corn crop have been identified. The next step is to consider how those nutrient needs can be met. Continue exploring the fertilizer planning process by looking at fertilizer options.
Fertilizer Options
Many sources of fertilizer are available to producers. These sources often include organic and inorganic forms. Two main sources are manure and commercial products.
Manure Sources
Manure provides many of the nutrients needed to raise crops, making it an ideal source of fertilizer. The amount of nutrients within manure can vary by the livestock source, whether that source is from a dairy, beef, poultry, or even a swine operation. It can also vary by the nutrient content of the feed being fed to those sources. Dairy cows being fed certain feed mixes or rations can have significantly different amounts of nutrients available in their manure compared with dairy cows receiving other feed mixes or rations.
Test manure samples to determine accurate nutrient levels before applying manure to any fields. The remaining nutrients not provided in the manure can then be met using commercial products.
Commercial products often have a greater availability than that of manure. There is also more uniformity in application as nutrient levels tend to remain relatively the same from one load to the next. However, commercial products are typically more expensive than manure alternatives. These considerations keep both options viable to producers depending on their situation.
Manure is highly regulated within Michigan because of the concern of overapplication and the desire to maintain safe levels of phosphorus in the soil.
To apply manure, you should follow the Generally Accepted Agricultural and Management Practices guidelines (GAAMPs) from the Michigan Right to Farm program (https://www.michigan.gov/mdard/0,4610,7-125-1599_1605---,00.html). These guidelines use the field’s soil tests to determine how much manure can be applied to a field (see Figure 5).
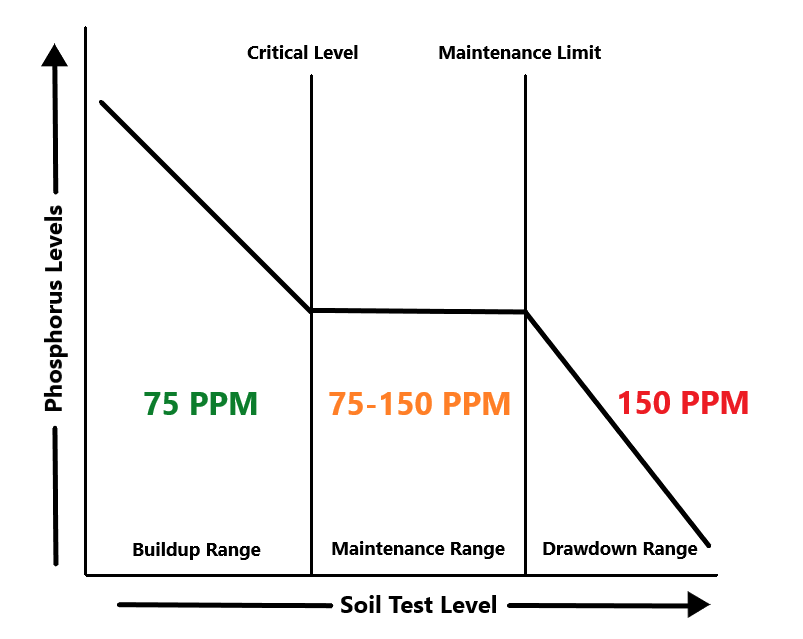
If soil phosphorus levels are higher than 150 ppm (equivalent to 300 lbs), you will need to draw down the nutrient levels before you apply more manure. If the test levels are below 75 ppm, you can apply manure to supply all crop nutrient needs.
If test levels show phosphorus is between 75 and 150 ppm, you can apply crop removal or maintenance amounts each year. However, while applications each year are an option, doing so is often impractical. One load of manure typically carries more than one year’s worth of crop removal. In an effort to recognize this, up to four years’ worth of crop removal can be applied at one time. Afterward, you’ll have to wait those four years before re-applying.
Commercial Products
Commercial products are regulated by state and federal laws as well. These products are required to display their nutrient content within their label.
At least three numbers are associated with the nutrient content in a label. The first number is the amount of nitrogen in the product; the second number, the phosphorus amount; and the third number, the potassium amount. This is often referred to as the NPK ratio and indicates the percentage of each nutrient regardless of whether the product is in a dry or liquid formulation. Nutrient ratios that have more than the NPK will always include the number and the letter corresponding to the nutrient such as the 17% sulfur included with potassium thiosulfate.
With dry products, determining the amount of nutrients being purchased is relatively easy to calculate. Diammonium phosphate (DAP) contains 18% nitrogen and 46% phosphorus in the form of P2O5 (see Figure 6).

Using 100 lbs of this product contains 46 lbs of P2O5. In that same 100 lbs, you will also receive 18 lbs of nitrogen.
How does this convert to tons if you are purchasing a ton of this product? A ton equals 2,000 lbs, therefore 46% of one ton of DAP contains 920 lbs of P2O5. A ton also contains 18% or 360 lbs of nitrogen.
For a liquid product, the focus is on gallons per acre being applied. This requires more math than using dry products because tons must be converted to gallons. To convert tons into gallons, the density of the product is a factor. Density refers to how much a gallon of the product weighs.
In the case of ammonium polyphosphate, or 10-34-0, each gallon weighs 11.7 lbs. The label indicates that for every pound there is 34% phosphorus in the form of P2O5. Multiplying 11.7 by 34% reveals that in every gallon of 10-34-0, there are 3.98 lbs of P2O5.
Ammonium polyphosphate also contains 10% nitrogen. Using the same density weight of 11.7 lbs and multiplying it by 10%, each gallon of the product contains 1.17 lbs of nitrogen (see Figure 7).

This product may also be purchased in tons. One ton contains 171 gallons (2000 lbs ÷ 11.7 lbs = 171 gallons) of product. If one gallon has 3.98 lbs of P2O5, then 171 gallons will have 680 lbs of P2O5 (3.98 lbs x 171 gallons). One gallon also has 1.17 lbs of nitrogen or 200 lbs in one ton.
Fertilizer Application Timing
The next consideration is how the fertilizer products will be applied. With most field crops, there are three main application periods: pre-plant, at the time of planting, or post-emergence.
The pre-planting period occurs before the seeds are planted in the soil. Typically, fertilizer is applied through broadcast spreading, incorporation into the soil, or both. Incorporation can be by tillage following broadcast or through injection.
The second method occurs at the time of planting, typically through the planter and often referred to as starter. The use of the term starter comes from the planter placing the fertilizer close to the seed. The placement of the fertilizer encourages more rapid and uniform growth in the field as the plant “starts” out (see Figure 8).

The last method is post-emergence, which occurs after the plants have emerged from the soil. This is accomplished by either broadcasting, spraying with pesticides, “Y” drops, or incorporation. These are often referred to as side-dress applications (see Figure 9).

As you think through the application strategies, consider their impact to the growing plants. If all fertilizer is applied at pre-plant, it may be too early for the crop to fully utilize. For example, there is a risk that fertilizer may leach or not be as readily available later in the season. Alternatively, if all fertilizer is applied at post-emergence, the crop may not grow uniformly during the season prior to the application. The crop could also suffer through nutrient deficiencies before the fertilizer is applied. Therefore, the costs of application timing can be just as impactful as the types of fertilizer being applied.
Consider an example where 250 lbs of nitrogen are applied at planting. Nitrogen leaching causes 30% or 75 lbs of lost nutrients over the course of the season. If these nutrients are valued at $0.55 per pound and multiplied by 75 lbs, then the loss to leaching is $42 per acre. In addition to the lost dollars per acre, a significant amount of nutrients is not available when the plant needs them later in the season.
Risks of nutrient availability and deficiencies are minimized by splitting the applications over multiple periods. This is especially important for sandy soils with lower CEC levels due to their lower nutrient-holding capacity. Some producers also make use of nitrogen stabilizers to extend the availability of nutrients in season. For more information on stabilizer products, review University of Nebraska–Lincoln’s Crop Watch article “Nitrogen Fertilizer Stabilizers in Corn” (https://cropwatch.unl.edu/2018/nitrogen-fertilizer-stabilizers-corn).
Type of Fertilizer Products to Apply
Deciding which type of products to use depends on the type of equipment, amount of available labor hours, and cost of nutrient products per application. For example, if your farm’s equipment is set up for dry fertilizer products, the use of any liquid products will require the purchase of new equipment or hiring of a custom applicator. Single-operator farms with no employees have a limited amount of time that they can spend on applications. This often requires custom hiring to assist in fertilizer applications and can present opportunities for both liquid and dry products to be used. These opportunities also extend to spreading of manure, especially if the farm does not own any livestock.
Creating a Fertilizer Plan
Everything to this point has been about understanding the components needed to create a fertilizer plan. Now it’s time to take that information and begin building the plan for your farm.
To help you think about this process, continue with the example corn farm with a yield goal of 150 bushels per acre. Imagine that the soil information indicates that the farm should focus on covering the crop removal amounts. Table 2 showed the farm will need 135 lbs of nitrogen, 55 lbs of phosphorus, and 40 lbs of potassium. These nutrient amounts maintain the soil at current levels and meet the yield goal.
Fertilizer Plan (Dry Products Only)
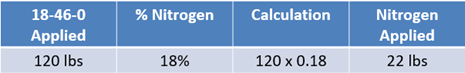
To apply crop removal needs using only dry products, first calculate phosphorus needs. Some fertilizer products containing phosphorus may also contain other nutrients, such as nitrogen. For example, diammonium phosphate (DAP) contains both phosphorus and nitrogen. When calculating how much of this product to use for phosphorus, a portion of nitrogen needs may be covered as well.
As you recall, 100 lbs of diammonium phosphate contains 46% or 46 lbs of P2O5 and 55 lbs of P2O5 are needed. If you divide 55 lbs by 46% the results indicate that 120 lbs of diammonium phosphate can be applied to meet the phosphorus needs (See Equation 1).

Now, consider the nitrogen being applied with the same product. There are 120 lbs being applied so how should nitrogen be considered? First, because of the concern of leaching over time, it is important to determine if the timing of the application will make the nitrogen available to the crop. If nitrogen will not be available, then it should not be counted toward meeting the yield goal. This is true of any nutrient that has a concern of leaching.
In this example, consider that all nutrients can be utilized and counted toward the yield goal. Since DAP contains 18% nitrogen in every pound, then the farm is receiving 22 lbs of nitrogen in the 120 lbs being applied (see Equation 2). Those 22 lbs can be subtracted from the overall nitrogen needs of 135 lbs to leave 113 lbs remaining.
To apply the remaining 113 lbs of nitrogen, the farm will be using urea. Urea is 46% nitrogen, which means every 100 lbs of the product contains 46 lbs of nitrogen. To find the urea amount to apply, divide 113 lbs by 46%. This calculation reveals that 246 lbs of urea are needed to reach the 150 bushels per acre yield goal (see Equation 3).
![]()
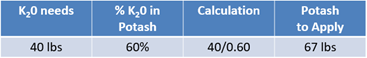
Now, consider potassium needs. The yield goal indicates that 40 lbs of K2O will be needed. Potassium chloride, or potash, will be used to achieve this goal. Potash or 0-0-60 contains 60% K2O in every pound of the product, which means that for every 100 lbs, there are 60 lbs of K2O. Divide 40 lbs by 60% and the results reveal that 67 lbs of potassium chloride should be applied (see Equation 4).
Remember that the methods used to apply these products will vary depending on equipment and timing available to the individual producer. The key is that the methods used should apply the nutrients at a time when they will benefit the crop. As discussed earlier, if you are applying a product such as diammonium phosphate in a pre-plant situation, you need to consider whether the nitrogen being applied will be of any benefit to the crop. In which case, you may want to re-evaluate which products to use to meet your nitrogen needs.
Fertilizer Plan (Liquid Products Only)
A fertilizer plan using dry product only was just developed. Now, consider using only liquid products and the same example of corn crop. Again, the farm needs to meet the nutrient removal amounts to reach its yield goal of 150 bushels per acre.
Just as with the dry products, start with phosphorus needs. In this case, use ammonium polyphosphate or 10-34-0 to supply the nutrients. As mentioned earlier, liquid products focus on an application rate of gallons per acre, which means the density of the product is a factor.
For 10-34-0, the density is 11.7 lbs per gallon and there is 34% of P2O5 in every pound. Therefore, one gallon of 10-34-0 contains 3.98 lbs of P2O5. You will need 55 lbs of P2O5 applied to the soil. Dividing the 55 lbs by 3.98 lbs per gallon reveals that 14 gallons of 10-34-0 will need to be applied (see Equation 5).
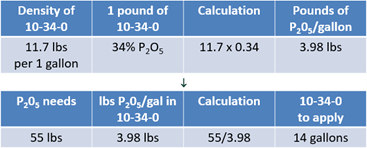
Now, shift to nitrogen needs, and 135 lbs will be removed by the yield goal. The farm is already applying 14 gallons of 10-34-0, and it contains both phosphorus and nitrogen. You know that one gallon of 10-34-0 has a density of 11.7 lbs. If one pound contains 10% nitrogen and is multiplied by the density of 11.7 lbs, then there are 1.17 lbs of nitrogen per every gallon. Therefore, in the 14 gallons already being applied, the crop will receive 16 lbs of nitrogen (see Equation 6). This reduces the 135 lbs needed to 119 lbs.
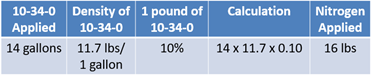
To apply the rest of the nitrogen, UAN in the formulation of 28-0-0 will be used. Some UAN products have different formulations, such as 32-0-0. For this example, the farm will use what is commonly called 28%.
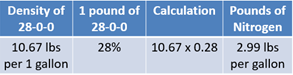
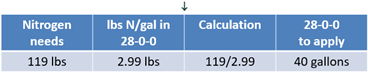
28-0-0 has a density of 10.67 lbs per gallon. Each pound contains 28% of nitrogen. Therefore, 10.67 lbs multiplied by 28% means every gallon contains 2.99 lbs of nitrogen (see Equation 7).
Dividing 119 lbs by 2.99 lbs reveals that the remaining nitrogen needs can be met by applying 40 gallons of UAN (see Equation 7).
Last, you need to apply 40 lbs of K20 to meet the potassium needs. To reach the yield goal, potassium thiosulfate (0-0-25-17S) will be used. This product has a density of 12.2 lbs per gallon and every pound contains 25% of K2O. Multiplying 12.2 lbs by 25% reveals that every gallon applied will provide 3.05 lbs of K2O (see Equation 8).
Dividing 40 lbs by 3.05 lbs of K20 indicates that 13 gallons should be applied to meet the potassium needs (see Equation 8).
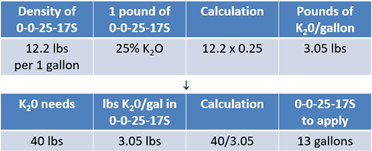
Like the dry products, these liquid products meet the nutrient removal needs set by the farm’s yield goal. A number of dry and liquid products can meet crop removal needs for your farm. Each offers their own sets of advantages and disadvantages.
Advantages & Disadvantages of Fertilizer Products
Dry products offer a higher versatility to mix and fine-tune applications, so more nutrients can be provided per application and reduce costs. Liquid products can be mixed but often require specific mixing directions to avoid chemical reactions that can be damaging to the crop. Dry products are often cheaper on a per acre basis.
However, liquid products have their own advantages. Plants prefer nutrients in forms that are easier to use. Nutrients in liquid products are typically in these desired forms, whereas many dry products require microorganisms to convert the nutrients. This helps to explain why liquid products are often used to correct nutrient deficiencies. They are taken up by the plant more quickly, which reduces potential yield loss. Liquid products are also easier to handle as they are less bulky and can be easily stored or transported in containers.
Most fertilizer plans use a mixture of liquid and dry products to reduce costs and maximize the benefits that each type of product offers. To illustrate this, consider the costs of fertilizer planning.
Costs of a Fertilizer Plan
Identifying the costs of a fertilizer plan involve several factors. These can include the cost of labor, fertilizer products, and product application. The cost of the products themselves tend to be the most variable, while costs for labor and product application are relatively more uniform across acres. Wages per hour for employees typically remain the same and applicators hired for custom work tend to charge a standard rate per acre. For more information on these custom rates, view the MSU Custom Machine and Work Rate Estimates factsheet at https://www.canr.msu.edu/resources/custom-machine-work-costs.
Manure costs will vary based on the source and the nutrient content expected from that source. The costs of commercial products can vary between the quantity of product purchased and the amount paid for them. Dry products are typically sold in tons, while liquid products can be sold in tons or by the gallon. A comparison of costs at each product’s application rates helps to determine which is more or less expensive.
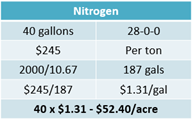
When liquid products are sold by the ton, a price conversion to gallons will match application rates. Achieve this by using the product density. For example, 28-0-0 UAN has a density of 10.67 lbs per gallon. 2,000 lbs divided by 10.67 equals 187 gallons. With a price of $245 per ton divided by 187 gallons, the price becomes $1.31 per gallon (see Equation 9).

To better understand variable costs of fertilizer, turn once more to the fertilizer plans for the example corn farm.
Cost of Fertilizer Plan (Dry Products)
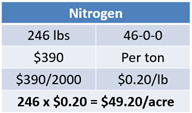
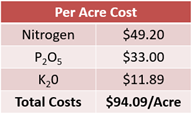
In the fertilizer plan with only dry products, nitrogen costs center around the 246 lbs of urea that will be applied. At a price of $390 per ton, this calculates out to $0.20 per pound. 246 pounds multiplied by $0.20 results in $49.20 per acre (see Equation 10).
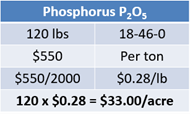
120 lbs of diammonium phosphate will be spread at a price of $550 per ton. This calculates out to $0.28 per pound. Multiply 120 lbs by $0.28 to find that the diammonium phosphate will cost $33.00 per acre (see Equation 11).
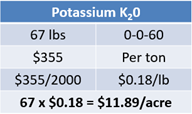
Lastly, potassium costs are based on using potassium chloride. Priced at $355 per ton, this calculates to $0.18 per pound or $11.89 per acre (see Equation 12).
Adding these costs together, the entire dry product fertilizer plan will cost $94.09 per acre (see Equation 13). How does this affect the overall revenue for the farm?
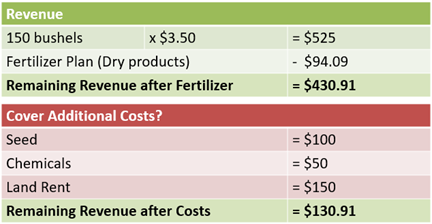
Considering the yield goal of 150 bushels and an estimated market price of $3.50 per bushel at harvest, the potential revenue is $525 per acre.
The fertilizer plan with dry products will cost $94.09 per acre, leaving $430.91 to cover additional costs. The additional costs would often include seed, chemicals, and rent, as well as other fixed and variable costs.
For example, if seed cost is $100 per acre, chemicals $50 per acre, and land rent is $150 per acre, this leaves $131 to cover repairs, fuel, insurance, and any overhead costs that remain (see Table 4).
Is $131 a reasonable amount of revenue to cover those expenses for the farm?
Is that enough remaining revenue to put toward any family living, real estate taxes, or debt payments that the farm would be expected to pay?
Cost of Nutrient Fertilizer (Liquid Products)
Now, look at the cost of the fertilizer plan with liquid products.
For nitrogen needs, 40 gallons of UAN (28-0-0) will be used at a price of $245 per ton. The tons need to be converted to gallons to find a price per acre. Using the density of the product and dividing by the pounds within a ton reveals there are 187 gallons per ton. Dividing the price of $245 per ton by 187 gallons equals a price per gallon of $1.31. Using the application amount of 40 gallons multiplied by $1.31 equals a cost of $52.40 per acre (see Equation 14).
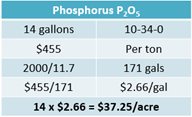
The farm plans to apply 14 gallons of ammonium polyphosphate (10-34-0) at a price of $455 per ton. There are 171 gallons per ton of this product. This means each gallon will cost $2.66. The cost of those 14 gallons is $37.25 per acre (see Equation 15).
Lastly, look at the potassium needs using potassium thiosulfate. The farm is applying 13 gallons at a cost of $630 per ton. Using the density factor of 12.2 pounds per gallon, there are 164 gallons in each ton of this product. Dividing the price of $630 per ton by 164 gallons results in a cost of $3.84 per gallon. Multiply the 13 gallons by $3.84. This equals a per acre cost of $49.94 (see Equation 16).

Adding these costs together, using only liquid products will cost $139.59 per acre (see Equation 17).

How does the cost of the fertilizer plan affect the overall revenue for the farm?
The expected revenue remains the same as the previous example with only dry products. The yield goal of 150 bushels multiplied by an estimated $3.50 per bushel equals $525 in potential revenue.
The fertilizer plan with liquid products will cost $139.59 per acre, leaving $385.41 to cover additional costs. These additional costs of seed, chemicals, and land rent remain the same from the dry products example (see Table 5).
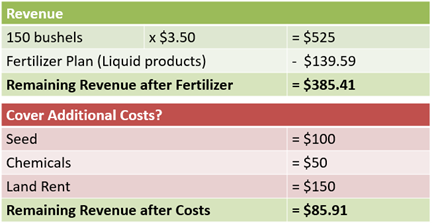
The same questions from the dry products example are also asked. Is $86 a reasonable amount to cover repairs, fuel, insurance, and any overhead costs that remain?
Is enough revenue remaining to cover costs of family living, real estate taxes, or debt?
As you consider the cost of these fertilizer plans, recall that using only dry products will cost $94.09 per acre. In comparison, only using liquid products will cost $139.59 per acre. This is a difference of $45.50 per acre between the two example plans.
This difference helps to illustrate why producers seek to use a mixture of both dry and liquid products. As the benefits of each product are considered, some nutrients can best be provided using dry products, while other nutrients by liquid products. By using both types of products, the cost of the fertilizer plan begins to shift somewhere between these two values and erode away the $45.50 per acre difference in the examples. Find the right combination of products that will meet the nutrient needs at the lowest possible cost to your farm.
Break-Evens of Fertilizer Plan
Another way to view the cost of these fertilizer plans is to consider them in a break-even position. The following focuses on a break-even for price and a break-even for yield.
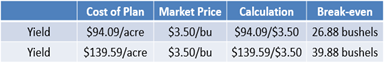
For the dry products, divide the cost of the plan by the yield goal of 150 bushels. This calculates a value of $0.63 per bushel needed to cover costs. Alternatively, if the farm considers a market price of $3.50 per bushel, it will need to sell 26.88 bushels at that price to cover the costs (see Equation 18).

For the liquid products, again look at both price and yield break-evens. For the price break-even, $0.93 per bushel is needed to cover costs. For break-even yield, the farm will need to sell 39.88 bushels to cover the costs (see Equation 19).

Take a closer look at the break-evens for both fertilizer plans. First, consider the break-even price and the $0.30 difference between dry and liquid programs (see Table 6). Understanding this comparison is where your role as a farm manager becomes important. As you think back to the advantages of liquid products, is $0.30 an acceptable cost for the benefits it offers? Or does the cost of the plan outweigh the benefits?

Consider the same questions in relation to the farm’s overall break-even. Does your answer change if the farm is using only dry products and its overall break-even is $3.40? If the market outlook is estimating $3.50 per bushel, how impactful would a change to only liquid products and increased costs of $0.30 per bushel be to the operation?
Use a similar thought process with break-even yield. In comparison, the dry fertilizer plan will need 13 bushels less than the liquid program to cover costs (see Table 7). With the advantages and disadvantages of dry products in mind, would saving the revenue of 13 bushels for other costs be best.
Does your answer change if production challenges (weather, available labor) are a concern?
Looking at break-evens for both price and yield illustrate the importance of understanding how much more a fertilizer plan will need from the marketing and production activities on the farm. By mixing both dry and liquid products in a plan, the extra burden on marketing and production activities is often reduced. These activities can include the extra cost of equipment or the need to outsource applications.
As you think about fertilizer planning, remember that detailed planning can help reduce the impacts to revenue. You can accomplish this by relying on accurate, routine soil testing to identify what nutrients are already present in the soil. Taking advantage of those available nutrients can reduce the amount of fertilizer to apply and still ensure that the plan meets your intended yield goals.
Most importantly, a detailed fertilizer plan helps to meet the nutrient needs of your crop at the lowest possible cost.



 Print
Print Email
Email