MSU investigating oak wilt as the cause of sudden chestnut tree decline
Prevention and a quick response are critical when it comes to oak wilt management.

In fall 2022, Michigan State University Extension personnel were called to a Michigan chestnut orchard to investigate the rapid collapse of 20-year-old ‘Colossal’ chestnut trees (Castenea sativa x C. crenata). Mature trees in one section of the orchard began to wilt in early June. Entire trees were affected, and tree death (rootstock and scion) occurred in less than two weeks. Wilt symptoms rapidly spread to neighboring trees in a radial pattern, affecting trees in the same row and in adjacent rows. The orchard and the surrounding area were visually evaluated for common causes of wilt in chestnuts, including chestnut blight, Armillaria, Phytophthora along with insect borers. Soil and chestnut tissue samples were collected and submitted for lab analysis. This initial orchard evaluation failed to identify any cause for the rapid collapse.
The expanding pocket of dead and dying trees matched the classic pattern of a wilt pathogen. To determine if a wilt pathogen was causing the issue, a fresh sample was collected from a branch of a declining, but still living tree. Importantly, the branch had both diseased and healthy-looking leaves on it. The Michigan State University Plant & Pest Diagnostics lab was able to detect a fungus called Bretziella fagacearum (Bf) from the branch material and subsequently grew the fungus in a culture. This fungus is the agent that causes a disease called oak wilt, which is found in 25 states in the U.S., and is present in at least 61 counties in Michigan.
Oak wilt affecting chestnut had not previously been confirmed or reported in Michigan. We identified a single report of the oak wilt fungus causing disease in Chinese chestnut (C. mollissima) in Missouri from the 1950s. However, we have since discovered that a chestnut grower in Ohio has lost more than a hundred 30-year-old trees with symptoms very similar to those seen in Michigan, and Bf was also isolated from these trees.
Given the apparent rapid decline and spread of oak wilt in chestnut, the potential impact could be severe in affected orchards. Luckily, although there is little published research about oak wilt and chestnut, there is substantial research on oak wilt in oak species. This work can help inform chestnut management while researchers work to better understand how the fungus behaves in chestnuts. If you suspect you have oak wilt in your chestnut orchard, please reach out to Erin Lizotte at taylo548@msu.edu.
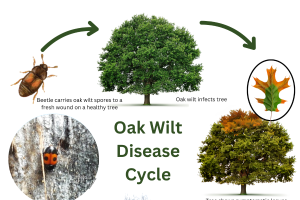
Disease cycle
Oak wilt primarily affects trees in the red oak group (Lobatae). Red oaks typically die within a year of infection. White oaks can be infected but generally do not succumb to the disease. Both oaks and chestnut are members of the family Fagaceae, so it is perhaps not surprising that oak wilt could infect chestnut trees. The following disease cycle is based on research with oaks but may shed some light on the behavior of oak wilt in chestnut.
Oak wilt can spread two ways. Overland spread occurs when tiny sap beetles, also known as picnic beetles (Family: Nitidulidae) pick up viable fungal spores from an oak that has recently died from oak wilt, then feed on sap oozing from a wound on a live oak. The wound must be deep enough to penetrate the sapwood.
When beetles move from a tree infected with oak wilt to a wound on a healthy tree, the spores can establish a new infection. The fungus grows into and eventually plugs up the xylem cells (sapwood) the tree uses to transport water. This causes the tree to wilt and die. Oaks infected in the spring can die within a few weeks. Trees infected in the fall may not die until the following year.
Beetles are most likely to be carrying spores in late spring and early summer. Therefore, activities that may wound trees, including pruning, should be avoided from April 15 through July 15 to reduce the risk of overland transmission and new oak wilt infections. Unavoidable or accidental wounds from storms or lawn equipment should be painted with a pruning sealer or latex paint as soon as possible.


Another way the disease spreads aboveground is through the movement of wood from oaks killed by oak wilt into new areas. Mycelial mats under the bark of a recently killed tree have a sweet odor that attracts sap beetles. When the tiny beetles visit the mat, they pick up the fungal spores that can then be introduced into a wound on a live tree. Mycelial mats can be produced on firewood or logs from trees killed by oak wilt during the previous year. Dry wood or completely debarked wood does not contain enough moisture for the oak wilt fungus to survive and is safe to move.
Once a tree is infected, below ground transmission of oak wilt from tree-to-tree can occur via root grafting between infected oaks and adjacent healthy trees. Root grafting, a natural phenomenon, occurs when adjacent or nearby trees of the same species form physical connections between their roots, allowing the trees to exchange water, carbon and nutrients. A tree killed by oak wilt can become the center of a disease outbreak, resulting in expanding pockets of dead and dying trees. When oak wilt is detected, management can involve disrupting the connected roots. This can be achieved by excavating deep trenches and removing infected oaks plus a buffer of healthy oaks to curtail disease spread.
Once oak trees are infected with oak wilt, they cannot be saved. Managing an outbreak early and effectively as described above can prevent additional oaks from becoming infected and dying.
Scouting

Growers may scout for oak wilt within their chestnut orchards and in any adjacent oak stands. Look for dead or declining trees. For reference, you may refer to the Michigan Department of Natural Resources (MIDNR) interactive map highlighting locations where oak wilt has been confirmed in Michigan. This map only shows detections officially reported to the MIDNR and is not a complete listing of all oak wilt locations.
An infected tree is often first noticed when leaves take on a wilted, light green appearance during the summer before progressing to yellow and brown and dropping prematurely. The presence of dark streaking under the bark on branches and between the cambium and bark on trunk cross sections are common symptoms of oak wilt. However, the only way oak wilt can be confirmed is either through observation of a mycelial mat forming under the bark or from laboratory testing in a diagnostic lab.
To date, mycelial mats have only been found on oak trees. Whether they will form on chestnut trees is not yet known. Growers who suspect they may have oak wilt affecting their chestnut tree may send a sample to MSU Plant and Pest Diagnostics following these guidelines.


Management
At this time, there are no published guidelines for oak wilt management in chestnut. Options for managing oak wilt in oaks, however, can provide a baseline management approach in chestnut. As research progresses, management tactics will be refined for chestnut orchard systems.
Prevention and a quick response are critical when it comes to oak wilt management. It is important to note that the sap beetles that vector the oak wilt are native, tiny and many species are present in the environment. It is not practical to prevent beetles from visiting wounded trees using insecticide treatments. The key to preventing oak wilt establishment is to avoid wounding trees from mid-April to mid-July to limit the risk of oak wilt transmission. This is when the mycelial mats are most abundant and contaminated sap beetles are most active.
Tools, clothing, shoes, equipment and soil do not spread the oak wilt fungus. The fungus, however, can form mats on wood from trees that were recently killed by oak wilt, including standing dead trees. Whether the oak wilt fungus can reproduce (i.e., form mycelial mats) in chestnut trees is unknown and therefore, we do not yet know if oak wilt-killed chestnut trees are a contamination source. To err on the side of caution, it is best not to store any material from oak wilt killed trees such as logs and firewood, near susceptible hosts like chestnut or oaks. Harvested trees from infected stands should either be immediately turned into lumber, burned or tightly covered by tarps (to prevent beetles visiting the wood).
In chestnut orchards where oak wilt is confirmed, growers may consider trenching to break apart any root grafts between trees to prevent belowground transmission of oak wilt. Maintaining the trench helps prevent further spread via root grafting by creating an air gap in which roots cannot grow and graft. Growers should create a trench around currently affected trees as well as their immediate healthy neighbors to help ensure the infection is contained.
Because the oak wilt fungus can spread through roots, and symptoms appear only after infection is established, an additional trench will need to be created around a buffer of apparently healthy trees encircling the diseased trees. While the trees may appear healthy, one or more of the trees may already be infected with oak wilt even though visible symptoms have not yet appeared.
If the root rupture/trenching efforts are deployed too close to affected trees, the fungus can continue to spread. For context, root grafting in hardwoods commonly spans gaps of 100 feet between trees. In oak trees, preventative applications of propiconazole injections may protect non-infected trees from the underground spread of the fungus if applications are re-applied every two to three years. Currently, it is not legal to inject propiconazole in edible chestnut orchards and there is no data on the effectiveness of injections in chestnut. There is no way to save a tree infected with the oak wilt fungus.
Additional resources
For more information on oak wilt, read "Worried about oak wilt?" from MSU Extension. For more information on sampling procedures for oak wilt testing, read "The best sampling procedures for accurate oak wilt testing" from MSU Extension.
This work is supported by the Crop Protection and Pest Management Program [grant no 2021-70006-35450] from the USDA National Institute of Food and Agriculture. Any opinions, findings, conclusions, or recommendations expressed in this publication are those of the author(s) and do not necessarily reflect the view of the U.S. Department of Agriculture.



 Print
Print Email
Email