Hydrolytic degradation of poly(lactic acid): Unraveling correlations between temperature and the three phase structures
, , , ,
Polymer Degradation & Stability, 2023, (217) 110537
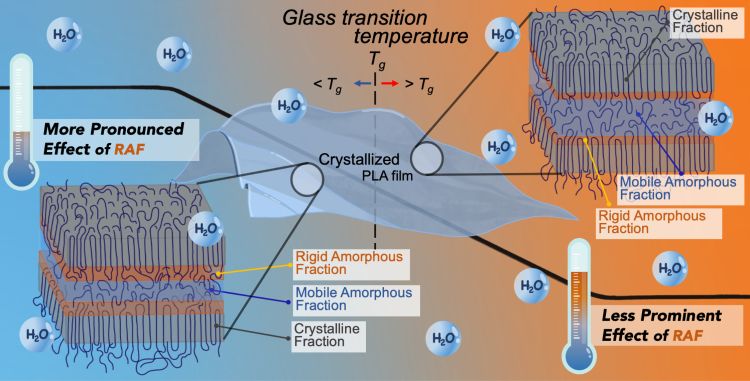
Hydrolysis significantly influences both the properties and degradability of poly(lactic acid), PLA. This work investigates the hydrolysis kinetics of PLA films as affected by degree of crystallinity and temperatures by considering the three-phase model structures (i.e., mobile amorphous, rigid amorphous, and crystalline). Molecular weight and three-phase fraction analyses were performed during hydrolysis to estimate the kinetic rates using phenomenological models. Results revealed that temperature significantly impacted PLA degradation, with distinct characteristics observed for each of these three phases. Above the glass transition temperature, the hydrolysis rates of PLA were comparable among samples with different crystallinity due to rapid water-induced crystallization of the amorphous phases, coupled with accelerated hydrolysis. In contrast, at below the glass transition temperature, the higher crystallinity sample exhibited a faster hydrolysis rate attributed to the presence of the rigid amorphous fraction. An increase in crystallinity introduced more defects due to limited mobility in the rigid amorphous fraction, influencing hydrolysis. The study provides valuable insights into the crucial relationship between temperature, crystallinity, and hydrolysis kinetics which are expected to be useful for predicting PLA degradation behavior during its intended applications and at its end of life.



 Print
Print Email
Email



