Pyrolysis to produce gasoline and biodiesel
Pyrolysis is a maturing technology that can yield infrastructure compatible fuels from renewable resources.
What is Pyrolysis?
There are several technologies and methods for converting plant biomass into a more useful energy product. Pyrolysis is a thermochemical process that heats (400 to 500 degrees Celsius) biomass in the absence of oxygen to create bio-oil that resembles crude oil.
To convert bio-oil to usable transportation fuels, the bio-oil is upgraded through hydrotreating and hydrocracking. In hydrotreating, hydrogen is reacted with the bio-oil in order to remove sulfur and oxygen. In hydrocracking, the hydrotreated bio-oil is reacted again with hydrogen in order to create smaller chains of hydrocarbons to meet the specifications of gasoline and diesel fuels (Hsu, 2012).
Environmental benefit
Life Cycle Analysis (LCA) is an accounting tool that is used to track inputs and outputs of processes and keep a running tally on specific traits. Here, LCA is used to track two items:
1) How much energy (Net Energy Value or NEV) is used to produce and deliver biomass, perform pyrolysis, upgrade the bio-oil and create gasoline and diesel fuel compared to the energy content of the fuel;
2) Carbon dioxide (a greenhouse gas) uptake by biomass and outputs from processing.
In both cases, a desirable outcome from pyrolysis of biomass would be to increase energy output and reduce CO2 emissions compared to fossil based alternatives.
The NEV (Figure 1) is 1.09 MJ km−1 for pyrolysis-derived gasoline and 0.92 MJ km−1 for pyrolysis-derived diesel, both higher than the NEV for conventional fossil-based gasoline of −1.2 MJ km−1. A positive number indicates that there is more energy in the fuel than is used to create it – or a positive gain in total energy. Note the negative number for fossil-based gasoline indicating a net loss of energy. The gain in energy for biomass-based fuels is a major reason why this technology is being considered.
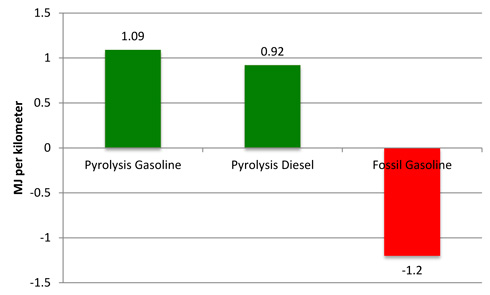
Figure 1.
Net Energy Value (NEV) of biomass based pyrolysis fuels compared
to
fossil-based gasoline. MJ=Megajoules is a measure of energy content.
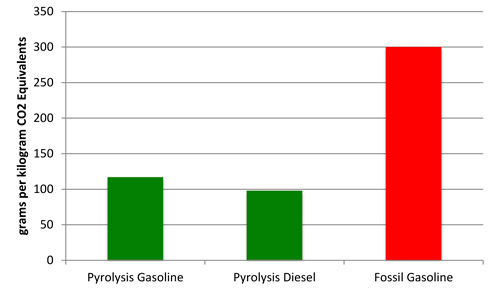
Figure 2.
Life Cycle Analysis of GHG emissions of pyrolysis based fuels
compared to
fossil-based gasoline.
Hsu (2012) calculated the greenhouse gas emissions of fuel derived from forest residues and determined that gasoline and diesel produced via pyrolysis are estimated to have greenhouse gas (GHG) emissions of CO2 equivalent (Figure 2) of 117 g km−1 and 98 g km−1, respectively, and about 65 percent lower than conventional gasoline (300 g km-1) (Argonne National Lab, 2012). Pyrolysis-based fuels from biomass have a clear advantage with less than half the amount of GHG emissions compared to fossil-based gasoline. If you look more closely at what is contributing to GHG emissions for pyrolysis fuels, you will find that grid electricity and natural gas account for 81 percent of the net GHG emissions. Utilizing electricity generated from biomass would further improve (reduce) the GHG emissions.
Next Steps
Evaluation of technology for production of renewable energy must meet a minimum litmus test to be considered as a contender for commercial scale production. Pyrolysis of biomass has passed the first two tests – net increase in energy output and reduction in GHG emissions. A third test is economics. The technology must be able to create an affordable fuel that is competitive with fossil-based fuels. Development of a supply chain for biomass is needed. How this supply chain develops and business models used will play a significant role in whether the litmus test for economics comes back positive. Additional research is needed to refine and optimize pyrolysis techniques for various types of biomass. Once this technology is fully in place, pyrolysis could be a significant contributor to meeting federal renewable energy goals.
You can follow progress in the bioeconomy at Michigan State University Extension’s website in the business section.
References
- The greenhouse gases, regulated emissions, and energy use in transportation (GREET) model, Argonne national Laboratory.
- David D. Hsu, Life cycle assessment of gasoline and diesel produced via fast pyrolysis and hydroprocessing, Biomass and Bioenergy, Volume 45, October 2012, Pages 41-47, ISSN 0961-9534, 10.1016/j.biombioe.2012.05.019.



 Print
Print Email
Email

