Bulletin E3488
Soil Microbial Community Dynamics Across Michigan Farms
February 26, 2024 - Benjamin Agyei, Michigan State University; Eric Anderson, Michigan State University Extension; Christina Curell, Michigan State University Extension; Maninder Singh, Michigan State University
Key Points
- Soil health differs significantly across Michigan regions.
- Soil texture did not account for major variations in soil health in this study.
- Tillage significantly impacts soil microbial community composition and overall soil health in Michigan.
- Increasing crop rotation diversity can enhance soil microbial community composition and improve overall soil health.

Introduction
Soil health is an important concept that has grown exponentially in the public and private sectors over the past decades. Soil health is defined as the capacity of soil to sustain plant and animal productivity, maintain and enhance soil and water quality, and support human health and wildlife habitat. Because several physical, chemical, and biological indicators exist within the concept of soil health, assessments and interpretations are complex. Additionally, the added expense of soil testing can prevent some growers from bothering to test at all. Growers who manage to test often go for basic soil test packages offered by soil-testing laboratories. These basic soil health packages mostly include soil pH, organic matter, available P, exchangeable Mg, K, Ca, and cation exchange capacity. A key indicator commonly not tested is microbial community. The soil microbial community plays critical roles, including nutrient cycling, carbon mineralization, and biological nitrogen fixation. Examples of some important groups of soil microbes and their functions are:
- Bacteria: Supply of plant nutrients, plant growth stimulation, plant pathogens inhibition, soil structure improvement
- Fungi: Organic matter decomposition, nutrient cycling, soil structure formation
- Arbuscular mycorrhizal fungi (AM fungi): Plant nutrition improvement, environmental stress tolerance and resistance, soil structure formation
- Actinomycetes: Nitrogen fixation, organic matter decomposition, phosphate solubilization
- Eukaryotes: Organic matter decomposition, soil structure formation, nutrient cycling
The soil microbial community is influenced by soil texture, vegetation cover, climate, and soil-crop management practices. In Michigan, regional differences among soil microbial communities are likely due to the large variability in cropping systems and weather conditions.
This report aims to:
- Highlight differences in soil microbial community structure across key production regions in Michigan.
- Describe how soil texture, tillage, and crop diversity affect the soil microbial community.
Overall, findings from this report will assist growers in adopting soil-crop management practices that promote soil health.
Methods
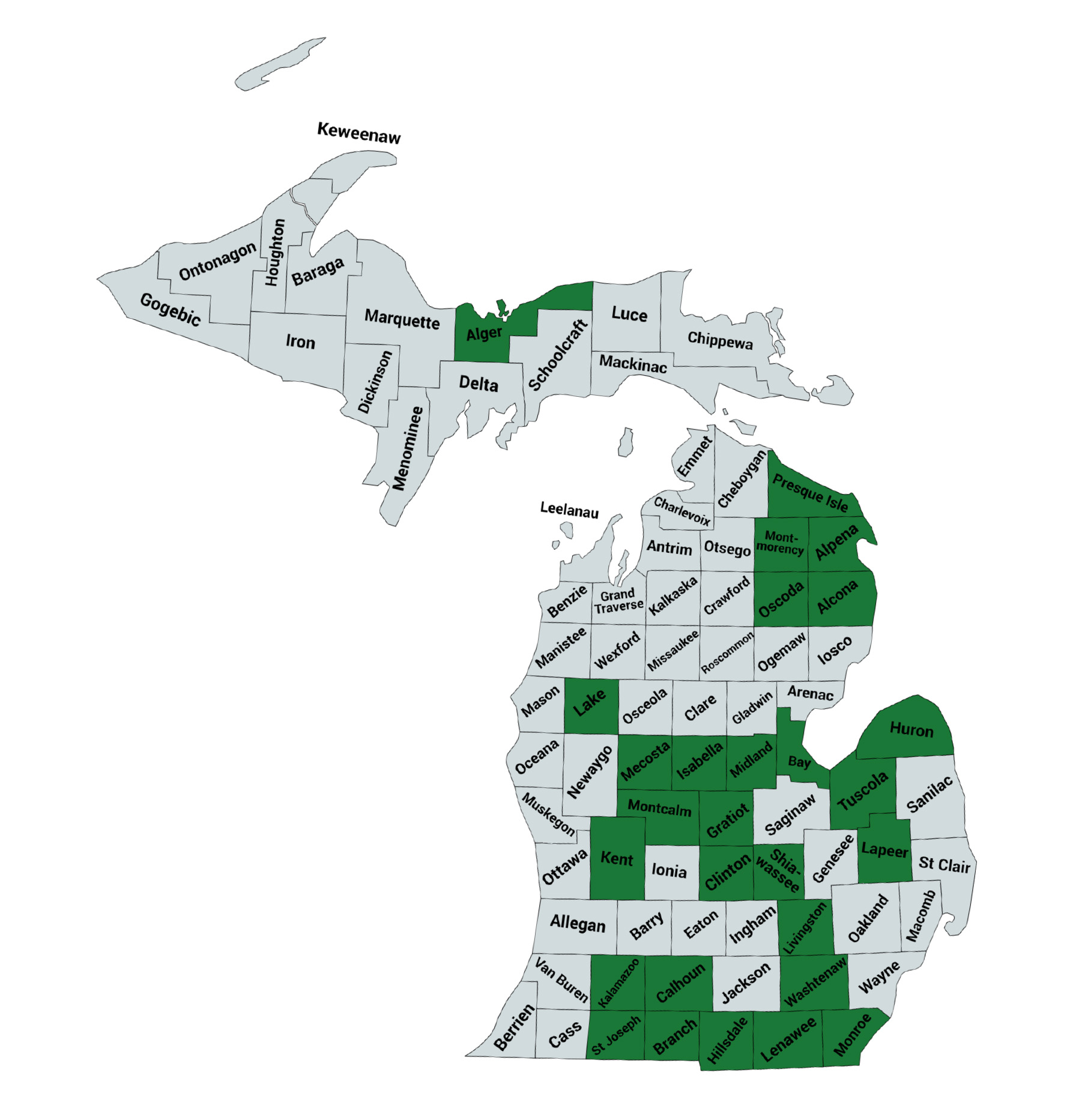
Soil sampling occurred fall 2019 through spring 2021. Seventy-four fields were sampled in fall 2019, 65 in spring 2020, 86 in fall 2020, and 85 in spring 2021, resulting in 310 samples. The samples came from 29 counties across Michigan (Figure 1).
The soil microbial community was analyzed using phospholipid fatty acid tests (PLFA) on samples each year at the University of Missouri soil health testing lab. The PLFA analysis provided the following output:
- Total microbial abundance: the sum of all the microbes present in the soil at the depth sampled (0-15 cm)
- Microbial community populations: the levels of the different kinds of microbes present in the soil
We evaluated five different microbial groups: arbuscular mycorrhizal (AM) fungi, bacteria, eukaryotes, fungi, and actinomycetes. These microbes are associated with several of the key soil processes and functions. Additionally, a field management survey form was sent to growers whose fields were sampled. Growers were asked to report crop rotation, tillage type, manure use, and cover crop use. Of the 310 samples, information on crop rotation and tillage was reported for 137 samples. Manure and cover crop use were reported for only five fields and were excluded from the final analysis.
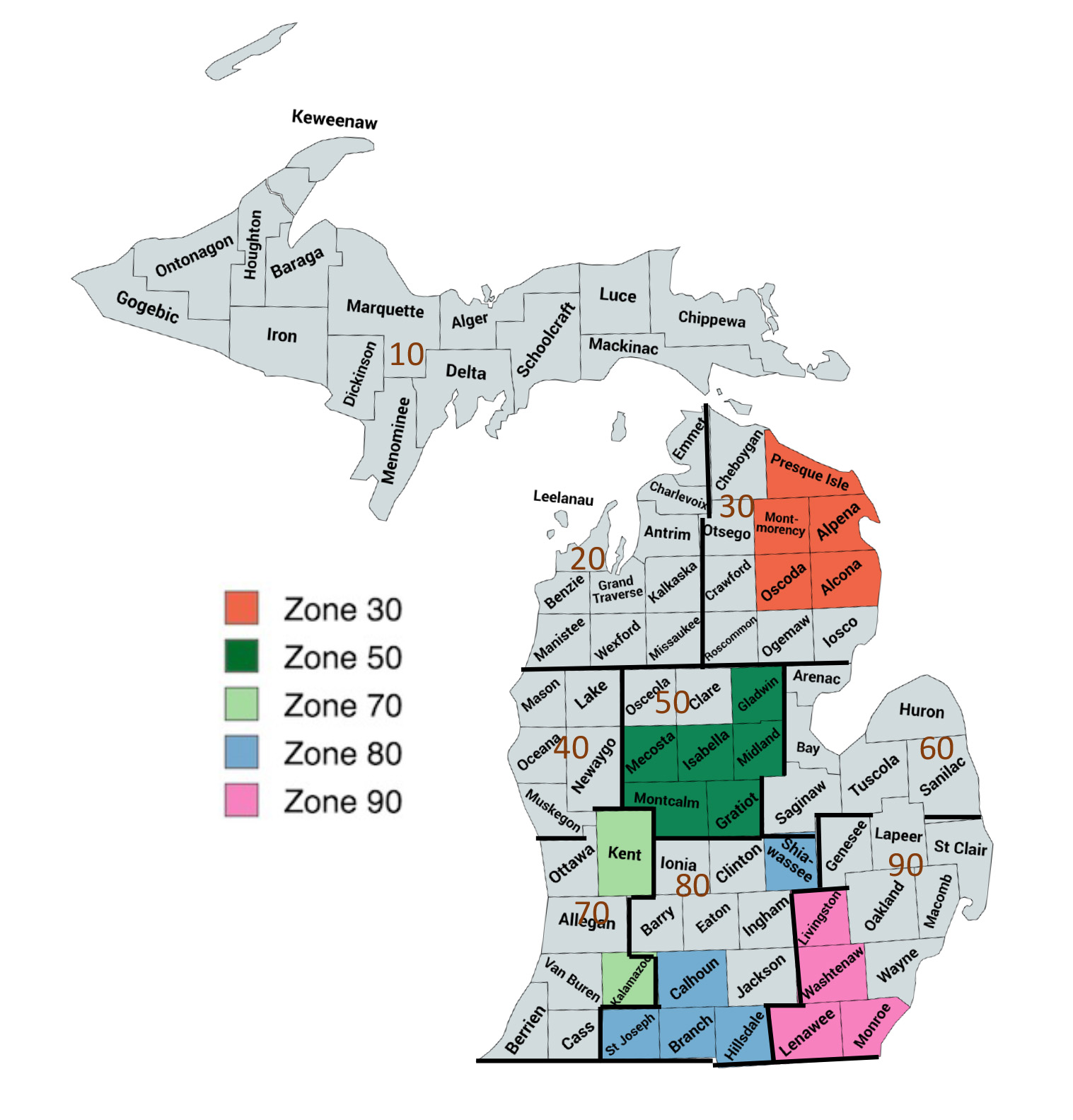
The data were first analyzed based on U.S. Department of Agriculture (USDA) crop districts. Five districts (30, 50, 70, 80, and 90), herein referred to as zones, were included in the final analysis (Figure 2). These zones occupy most of Southwest, Southeast, Central, and Northeast Michigan. Soil texture was classified first into 12 textural classes and further categorized into one of three main soil groups: coarse (sand, loamy sand, sandy loam), medium (sandy clay loam, loam, silt loam, silt), and fine (sandy clay, clay loam, silty clay loam, silty clay, clay) (Sprunger et al., 2021). The texture class was determined using the Web Soil Survey (https://websoilsurvey.nrcs.usda.gov/app/WebSoilSurvey.aspx).
The final analyses examined tillage and crop rotation. Fields that had undergone any form of tillage were classified as tilled while fields with no history of tillage were classified as no-till. The crop diversity index (CDI) was calculated as the total number of species in the three-year crop rotation before sampling. A total of three CDIs were estimated as follows: 1 (Corn-Corn-Corn), 2 (Corn-Soybean-Corn), and 3 (Corn-Soybean-Wheat or any other crop in the mix). Data analysis was conducted by fitting a linear mixed model and mean separations were done using Tukey’s test at p < 0.05.
Results
Microbial Abundance Across Zones
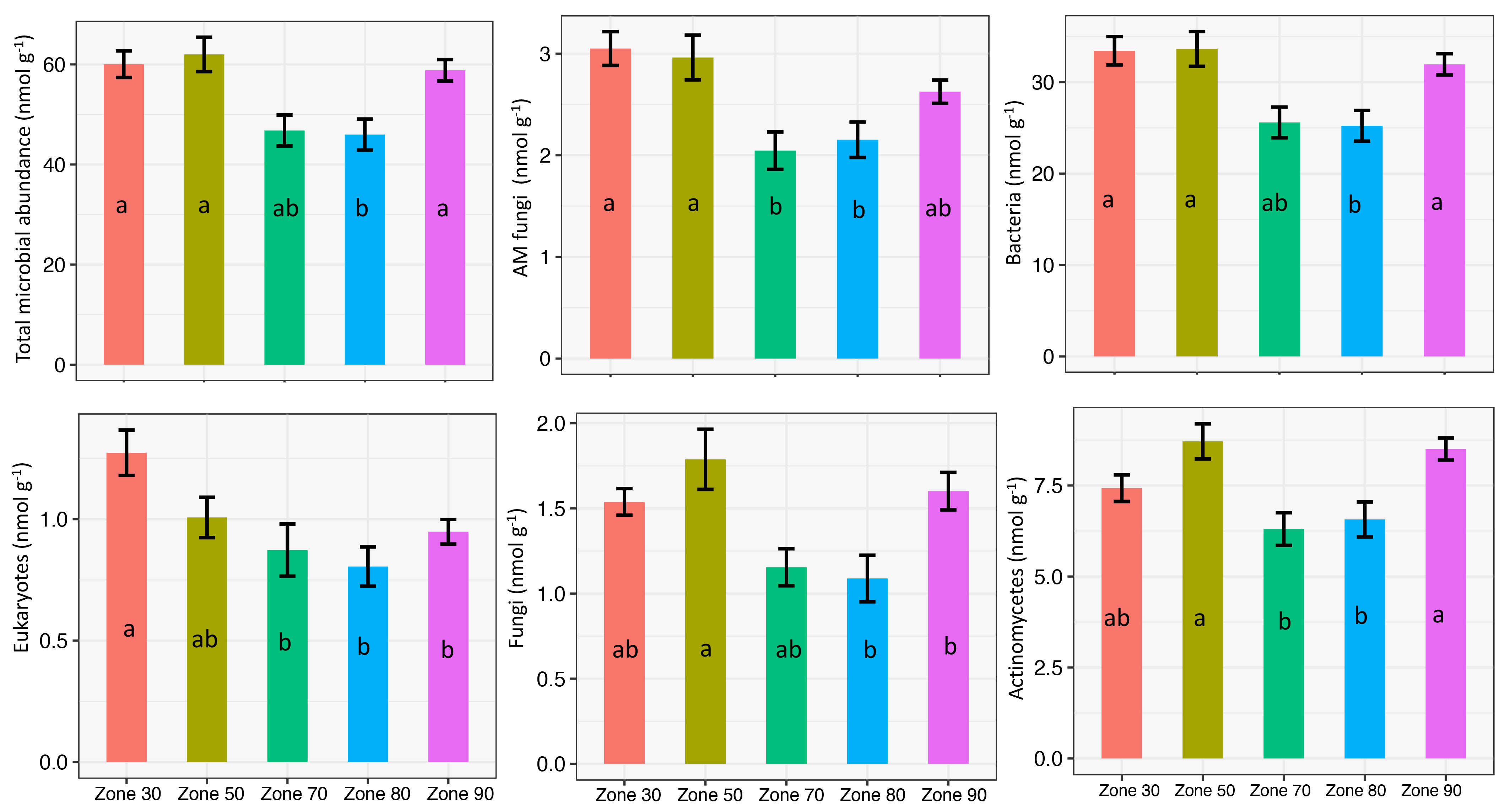
Overall, microbial abundance differed significantly across the five zones. Zones 30, 50, and 90 consistently had higher and similar microbial abundance, while zones 70 and 80 had significantly lower levels (Figure 3). The observed difference across the various zones could be due to variations in soils and agronomic management practices. In terms of soil texture, zones 70 and 80 had predominantly coarse soil texture with low clay fraction (10–25% clay). In terms of agronomic practices, most fields (>50%) in zones 70 and 80 were under conventional tillage.
Microbial Abundance Across Soil Texture Class
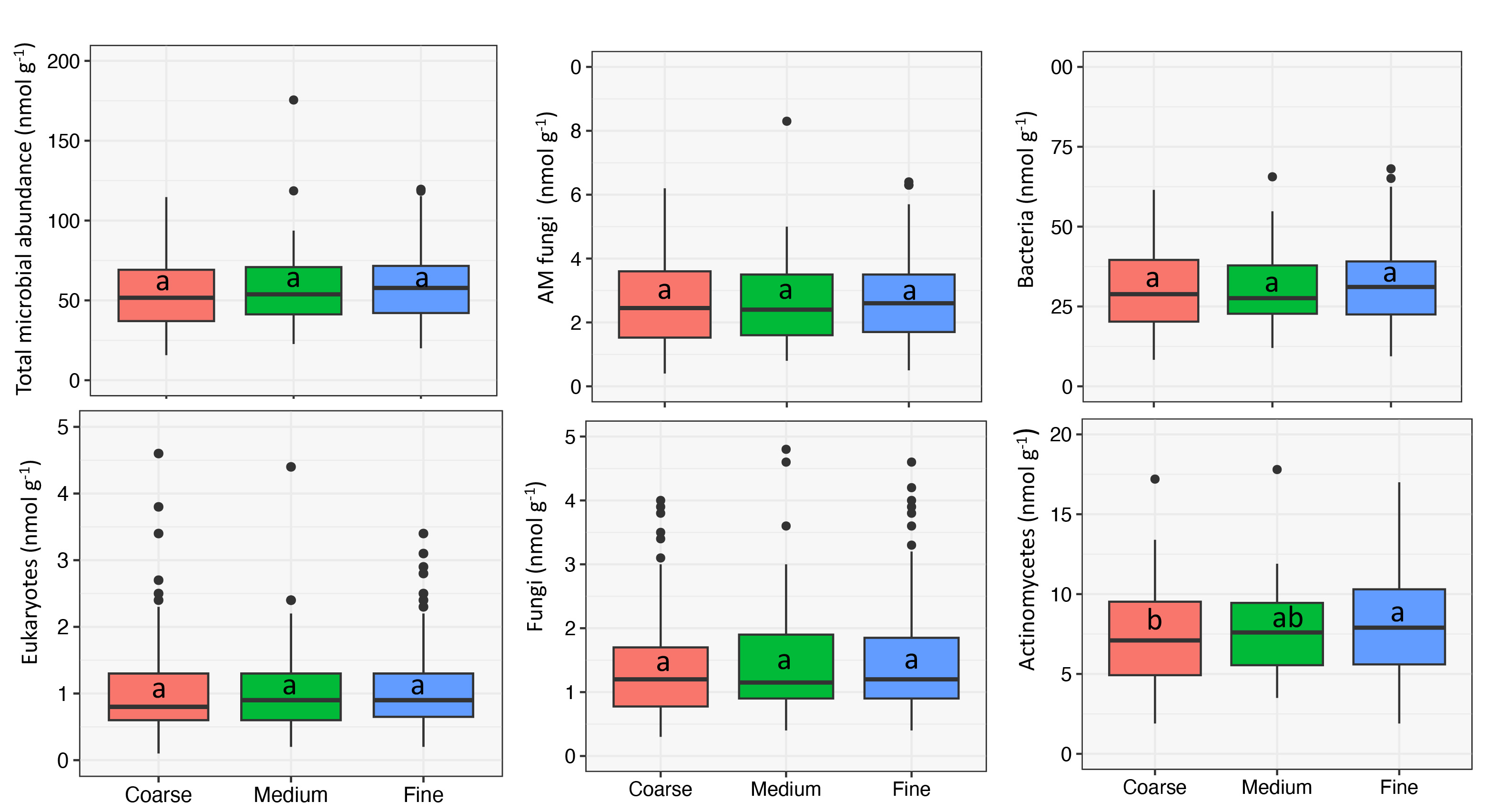
Overall, microbial abundance was similar across texture classes except for actinomycetes populations which was significantly higher in fine-textured soil (Figure 4). Additionally, we observed an increasing trend of higher bacterial and AM fungi populations among fine-textured soils with higher clay content. Even though we found no significant effect of texture on microbial abundance, it is important to mention that some studies have reported a significant effect of texture on soil microbial populations (Xia et al., 2020).
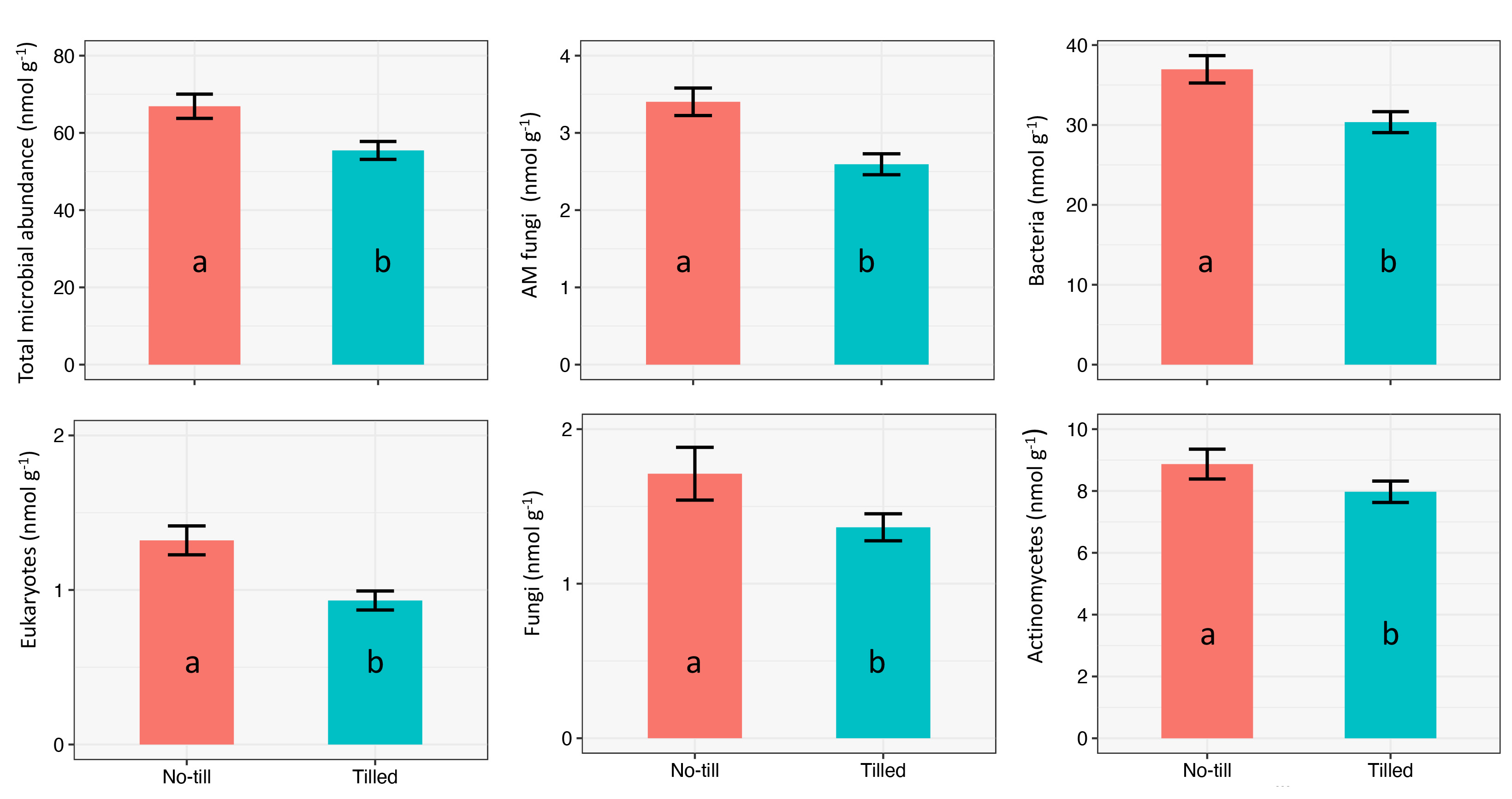
Microbial Abundance Across Tillage Type
Tillage significantly impacted microbial abundance (Figure 5). Overall, total microbial abundance was significantly higher under no-till than tilled. Additionally, bacteria, fungi, AM fungi, actinomycetes, and eukaryotes populations were significantly higher under no-till. Higher soil microbial populations are often associated with high soil carbon since carbon is a substrate for microbes. We examined total organic carbon and permanganate oxidizable carbon (POXC) and found significantly higher levels under no-till compared to tilled.
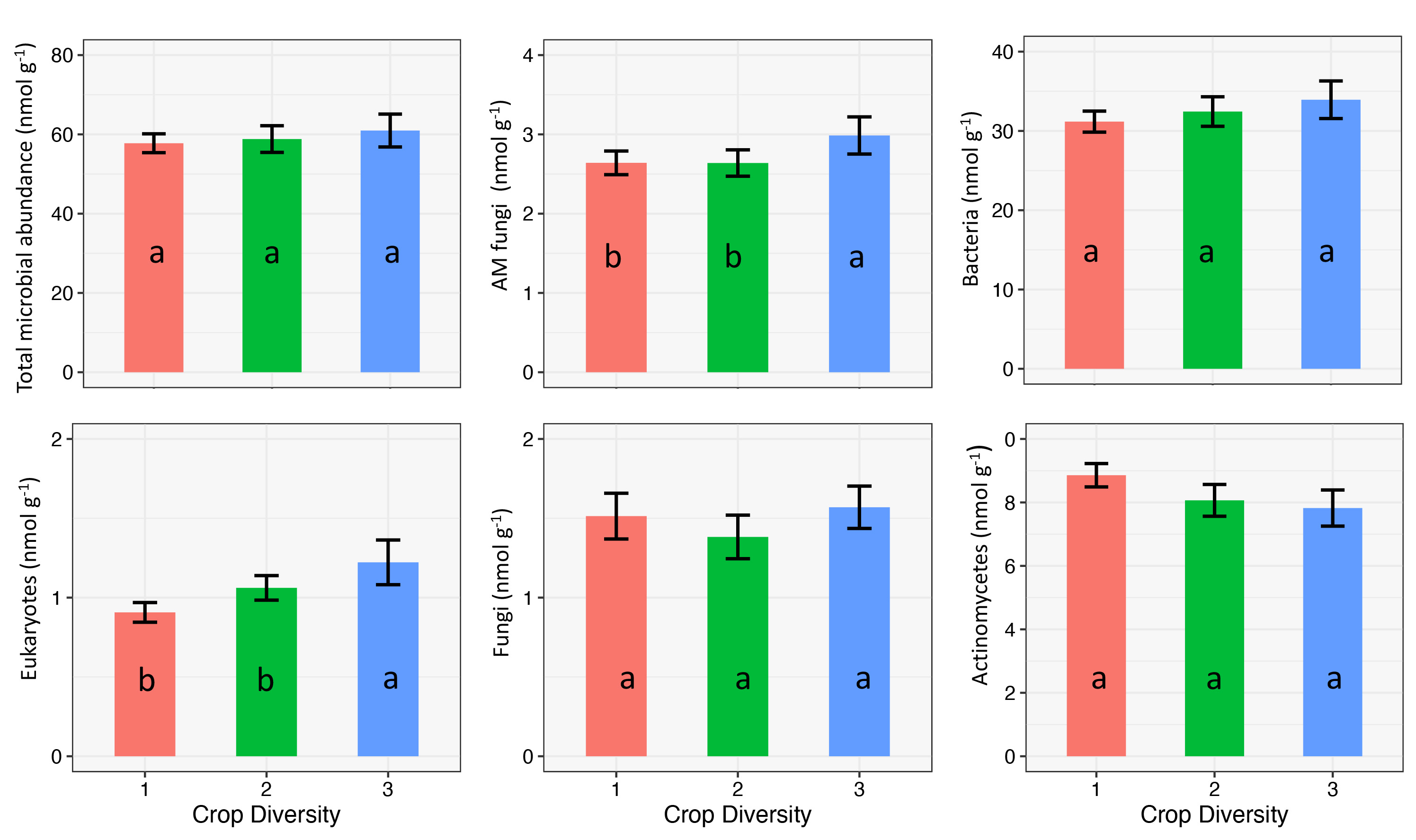
Microbial Abundance Across CDI
Overall, the total microbial abundance was mostly similar across CDI levels (Figure 6) except for AM fungi and eukaryotes populations, which were significantly higher at CDI of three. We also observed a trend of consistently higher bacteria and fungi populations with greater crop rotation diversity. The most common crops in the rotation were corn, soybean, wheat, dry bean, and alfalfa. Increasing crop diversity leads to greater residual crop biomass, litterfall, higher levels of carbon, and nutrient resources for soil microbes (Chen et al., 2019). These factors have a strong potential to increase microbial abundance in the soil.
Dataset Limitations in This Study
Our study provides some insight on how texture, tillage, and crop diversity affect soil microbial populations. Our study is unique in a way because of the high number of fields used, which is not typical in most on-farm studies. However, our dataset has some limitations. First, we had limited data in some regions; hence, we excluded them from final analyses. Second, we examined only three years of management information with a focus on only tillage practices and crop rotation. We did not examine other management factors such as manure amendment, residue management, field drainage, and cover crops. These management factors have the potential to affect soil microbial abundance and community composition.

Recommendations
Enhancing soil microbial abundance is key to improving overall soil health. Some studies (Culman et al., 2012; Hurisso et al., 2016) have reported a positive influence on crop yields with soil health management practices. Growers should adopt principles that have been identified to enhance soil health. Examples of key management principles based on literature and findings from this study include:
- Maximize presence of living roots:
- Plant cover crops
- Maximize biodiversity:
- Increase crop diversity
- Plant cover crops
- Integrate livestock
- Minimize disturbance:
- Reduce tillage frequency
- Reduce tillage passes
- Switch to no-till
- Maximize soil cover:
- Leave crop residues on soil surface
- Mulch
- Add compost
- Plant cover crops
Acknowledgments
Funding for this project was awarded by the Michigan Department of Agriculture and Rural Development under Fertilizer Research Grant #190000000603, “Benchmarking Cornstalk Nitrate-N, Soil P, and Soil Health Attributes on Diverse Cropping Systems.”
This bulletin is a condensed summary of this research project. To learn more about the study, read the article “Farm-Level Variability in Soil Biological Health Indicators in Michigan is Dependent on Management and Soil Properties” published in the Soil Science Society of America Journal (https://doi.org/10.1002/saj2.20630).
The authors would like to thank the following Michigan State University Extension staff for their contributions in working with farmer cooperators and data collection for this project:
- Bob Battel
- Ricardo Costa
- James DeDecker
- Paul Gross
- Jim Isleib
- Monica Jean
- Phil Kaatz
- Christian Tollini
References
Chen, C., H.Y.H. Chen, X. Chen, and Z. Huang. 2019. Meta-analysis shows positive effects of plant diversity on microbial biomass and respiration. Nat. Commun. 10(1): 1–10. doi: 10.1038/s41467-019-09258-y.
Culman, S.W., S.S. Snapp, M.A. Freeman, M.E. Schipanski, J. Beniston, et al. 2012. Permanganate Oxidizable Carbon Reflects a Processed Soil Fraction that is Sensitive to Management. Soil Sci. Soc. Am. J. 76(2): 494–504. doi: 10.2136/sssaj2011.0286.
Hurisso, T.T., S.W. Culman, W.R. Horwath, J. Wade, D. Cass, et al. 2016. Comparison of Permanganate-Oxidizable Carbon and Mineralizable Carbon for Assessment of Organic Matter Stabilization and Mineralization. Soil Sci. Soc. Am. J. 80(5): 1352–1364. doi: 10.2136/sssaj2016.04.0106.
Sprunger, C.D., S.W. Culman, L. Deiss, C. Brock, and D. Jackson-Smith. 2021. Which management practices influence soil health in Midwest organic corn systems? Agron. J. 113(5): 4201–4219. doi: 10.1002/agj2.20786.
Xia, J., R. Ren, Y. Chen, J. Sun, X. Zhao, et al. 2020. Multifractal characteristics of soil particle distribution under different vegetation types in the Yellow River Delta chenier of China. Geoderma 368(September 2019): 114311. doi: 10.1016/j.geoderma.2020.114311.



 Print
Print Email
Email






